EdU Imaging Kits (HF594)
Measuring cell proliferation and cell cycle are a fundamental method to assess cell health, determine genotoxicity, and evaluate drug’s pharmacodynamic effect. The common method is measuring DNA synthesis directly. In previous experiments, there are several approaches such as the incorporation of radioactive nucleosides (3H-thymidine) or BrdU. Here, we introduce one new method, click chemistry-CuAAC (Copper-Catalyzed Azide-Alkyne Cycloaddition), and the use of this reaction in direct measurement of S-phase DNA synthesis in cell cycle.
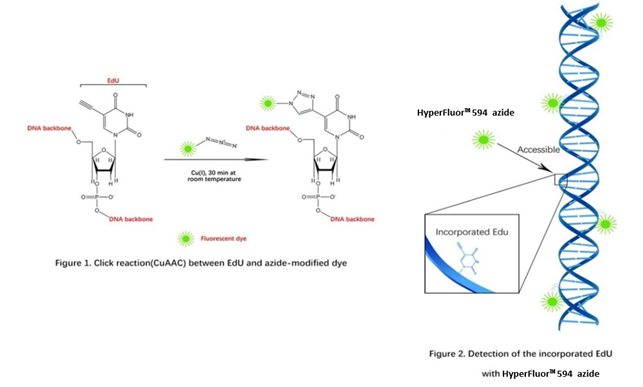
A nucleoside analog of thymidine, EdU (5-ethynyl-2’-deoxyuridine), can be incorporated into DNA strand during DNA synthesis. The alkynyl group of EdU is a biologically inert group that will undergo an extremely selective reaction with dye’s azido via a CuAAC reaction to afford an 1,2,3-triazole product. EdU and HyperFluor™ 594 azide possess biologically unique moieties to label DNA of proliferating cells, producing low backgrounds and high detection sensitivities. This CuAAC reaction affords superior regioselectivity and quantitative transformation under extremely mild conditions.
EdU Imaging Kits (HF594) specifically labels the DNA of proliferating cells after HyperFluor™ 594 azide is ligated to EdU, then the proliferating cells can be detected by a microscope or flow cytometry.

- 1. Ling Wang, Qicai Liu, et al. "Prostaglandin E2-driven dedifferentiation of Schwann cells leads to perineural invasion in pancreatic ductal adenocarcinoma." Signal Transduct Target Ther. 2026 Apr 7;11(1):122. PMID: 41946682
- 2. Jialong Deng, Bin Xu, et al. "Overcoming gefitinib resistance and metastasis in adenocarcinoma through synergistic PI3K–AKT–ERK blockade: Integrated rational co-delivery nanoplatform and …." Biomater Transl. 2026
- 3. Yan Hu, Chuntao Liu. "SIRT3-SUMO regulated Treg cell differentiation and asthma development by mediating N-glycosylation through the FAO pathway." Cell Biol Toxicol. 2025 Dec 13;41(1):164. PMID: 41388161
- 4. Yujie Ma, Runqiang Liu, et al. "Oleic acid inhibits the proliferation, invasion and migration of tongue squamous cell carcinoma cells under high glucose conditions through S100A9-HK2/PKM2-SOD2 axis." Tissue Cell. 2025 Jun 24:96:103026. PMID: 40582120
- 5. Yingjie Cai, Tong Guo, et al. "Alpha-Linolenic Acid from Zanthoxylum Seed Powder Regulates Fatty Acid Metabolism and Influences Meat Quality of Pekin Duck via the ADIPOQ/AMPK/CPT-1 Pathway." J Agric Food Chem. 2025 Jun 11;73(23):14651-14665. PMID: 40459020
- 6. Yaomin Chen, Bin Chen, et al. "SENP1 promotes deacetylation of isocitrate dehydrogenase 2 to inhibit ferroptosis of breast cancer via enhancing SIRT3 stability." Biotechnol Appl Biochem. 2025 Jun;72(3):805-824. PMID: 39690748
- 7. Tingting Qin, Zhangxu He, et al. "Taohe Chengqi decoction improves diabetic cognitive dysfunction by alleviating neural stem cell senescence through HIF1α-driven metabolic signaling." Phytomedicine. 2024 Dec:135:156219. PMID: 39520950
- 8. Ziqiao Yuan, Jiahui Meng, et al. "Formononetin Mitigates Liver Fibrosis via Promoting Hepatic Stellate Cell Senescence and Inhibiting EZH2/YAP Axis." J Agric Food Chem. 2024 Oct 3
| Components | 50-500 Tests | |
| EdU (Component A) | 5 mg | |
| HyperFluor™ 594 azide (Component B) | 1 vial | |
| DMSO (Component C) | 4 mL | |
| 10X EdU Reaction Buffer (Component D) | 4 mL | |
| CuSO4 (100 mM Aqueous Solution) (Component E) | 1 vial | |
| EdU Buffer Additive (Component F) | 400 mg | |
| Hoechst 33342 (10 mg/mL in Water) (Component G) | 35 μL | |
Store at -20ºC, keep dry and protected from light, stable for 1 year. | ||








