ADC Linker
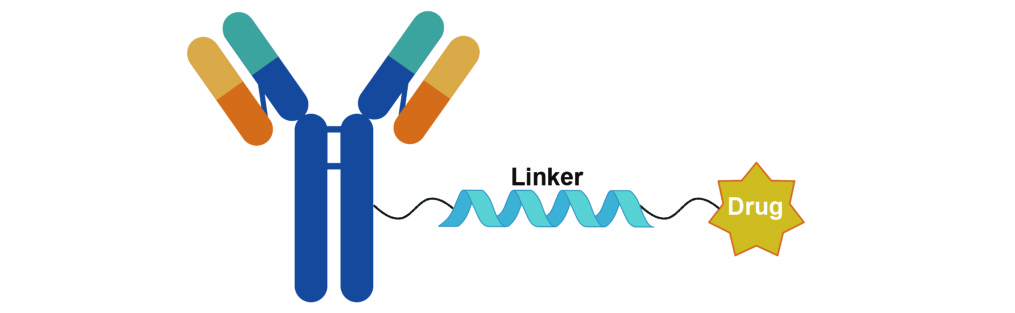

Antibody–drug conjugate (ADC) linkers are core functional components of ADCs, serving as covalent bridges that connect targeting antibodies to cytotoxic payloads. Their design is governed by the dual requirement of circulatory stability and efficient intracellular release. By employing specific chemical structures (such as peptide bonds, disulfide bonds, and acid-labile linkages), linkers enable precise control over ADC behavior: they preserve the integrity of the conjugate in the bloodstream to prevent premature payload release and off-target toxicity, while, after cellular uptake (e.g., via endocytosis and subsequent lysosomal degradation), they facilitate efficient payload liberation through enzymatic hydrolysis, reductive cleavage, or pH-responsive mechanisms. In this way, linkers form the molecular basis for both the targeting selectivity and safety profile of ADCs.
As essential reagents for ADC research and development, structurally diverse linkers provide critical support for both basic research and translational drug discovery. Different linker types (including cleavable, non-cleavable, and hydrophilically modified linkers) can be tailored to a wide range of payloads, such as microtubule inhibitors and DNA-damaging agents, thereby accommodating diverse molecular targets and heterogeneous tumor microenvironments. Linker stability, intracellular release efficiency, and biocompatibility directly shape the therapeutic window of ADCs and contribute to overcoming the resistance and toxicity limitations associated with conventional chemotherapy. In target validation, structure–activity relationship studies of ADCs, and preclinical drug screening, linker reagents thus provide crucial technical leverage for the precise modulation of ADC performance and for expanding the therapeutic applications of ADCs in oncology.
-
 BA2344 DRF-1042Summary: DRF-1042 is an orally active camptothecin analog with inhibitory effects on DNA topoisomerase I.
BA2344 DRF-1042Summary: DRF-1042 is an orally active camptothecin analog with inhibitory effects on DNA topoisomerase I. -
 BA2349 Gly-7-MAD-MDCPTSummary: Gly-7-MAD-MDCPT is an anticancer agent.
BA2349 Gly-7-MAD-MDCPTSummary: Gly-7-MAD-MDCPT is an anticancer agent. -
 BA2351 TH1338Summary: TH1338, an orally active derivative of camptothecin, is a promising anticancer compound that exhibits excellent tumor cytotoxicity in vitro.
BA2351 TH1338Summary: TH1338, an orally active derivative of camptothecin, is a promising anticancer compound that exhibits excellent tumor cytotoxicity in vitro. -
 BA2352 Ac-ExatecanSummary: Ac-Exatecan is acetylated modified.
BA2352 Ac-ExatecanSummary: Ac-Exatecan is acetylated modified. -
 BA2353 Exatecan-amide-cyclopropanolSummary: Exatecan-amide-cyclopropanol is an anticancer agent.
BA2353 Exatecan-amide-cyclopropanolSummary: Exatecan-amide-cyclopropanol is an anticancer agent. -
 BA2357 Decyclohexanamine-ExatecanSummary: Derivatives of camptothecin.
BA2357 Decyclohexanamine-ExatecanSummary: Derivatives of camptothecin. -
 BA2360 MensacarcinSummary: Mensacarcin is a highly complex polyketide that is commonly found in tumor cell lines to strongly inhibit cell growth and may induce apoptosis in melanoma cells.
BA2360 MensacarcinSummary: Mensacarcin is a highly complex polyketide that is commonly found in tumor cell lines to strongly inhibit cell growth and may induce apoptosis in melanoma cells. -
 BA2364 SMCCSummary: SMCC is a protein cross-linking agent.
BA2364 SMCCSummary: SMCC is a protein cross-linking agent. -
 BA2366 MC-Val-Cit-PABSummary: MC-Val-Cit-PAB is a synthesized intermediate and is a toxin-Linker coupling (Drug-Linker Conjugates for ADC).
BA2366 MC-Val-Cit-PABSummary: MC-Val-Cit-PAB is a synthesized intermediate and is a toxin-Linker coupling (Drug-Linker Conjugates for ADC). -
 BA2367 DBCO-amineSummary: DBCO-amine is a degradable (cleavable) linker for the synthesis of antibody coupled active molecules (ADCs).
BA2367 DBCO-amineSummary: DBCO-amine is a degradable (cleavable) linker for the synthesis of antibody coupled active molecules (ADCs).


