GPCR/G protein
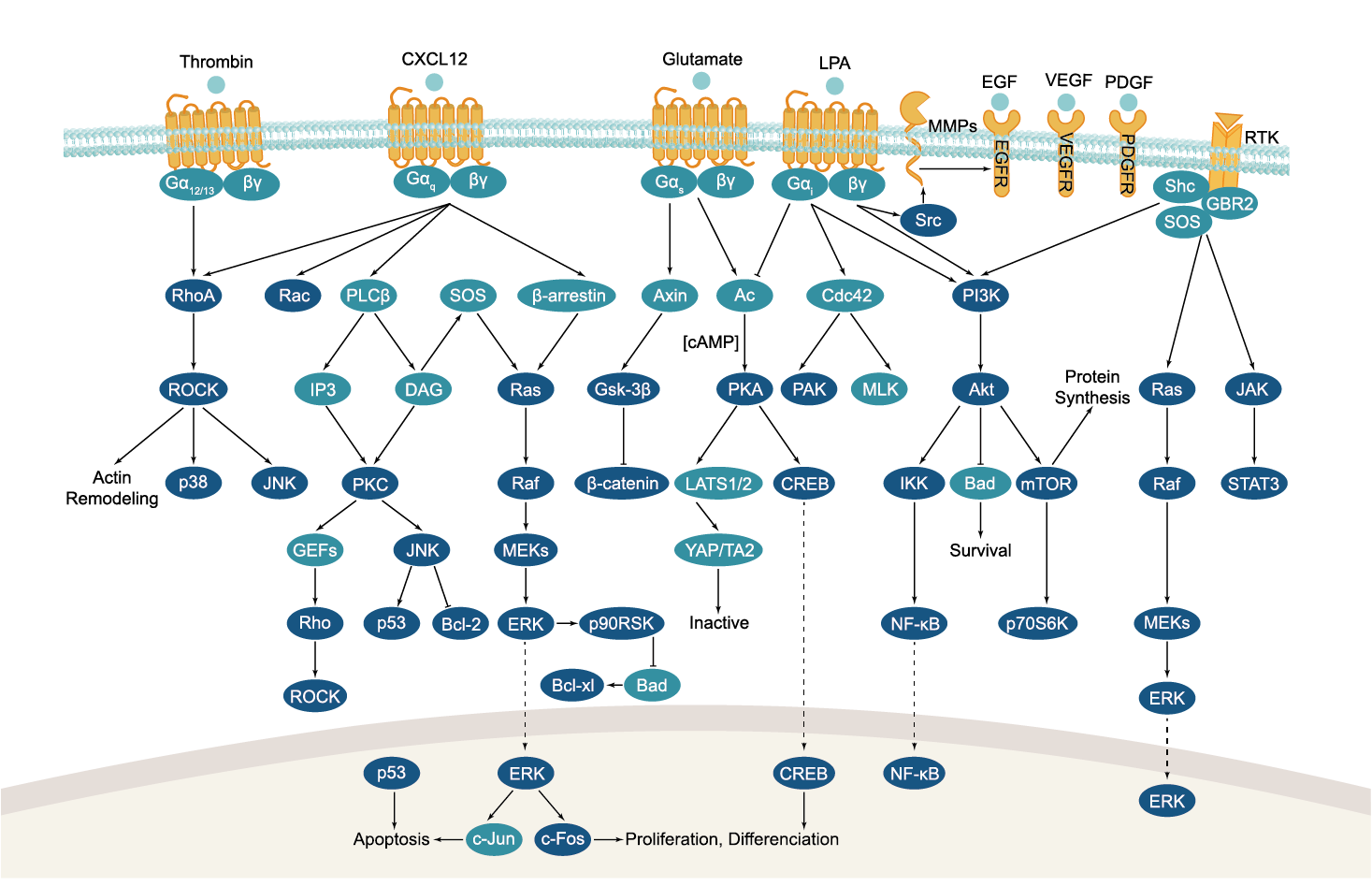

All GPCRs share a common seven trans-membrane structure. GPCRs are associated with heterotrimeric G-proteins which are GTP-binding proteins made of alpha, beta, and gamma subunits. When a ligand binds to GPCR, it activates the attached G-protein, the GDP is replaced with GTP. The activated G-protein then dissociates into an alpha and a beta-gamma complex which activates downstream signaling pathways. These intracellular signaling pathways include cAMP/PKA, calcium/NFAT, phospholipase C, protein tyrosine kinases, MAP kinases, PI-3-kinase, nitric oxide/cGMP, Rho, and JAK/STAT.
GPCRs are one of the most important therapeutic targets for various diseases, over 30% of all modern medicinal drugs target this family. Aberrant GPCR functions are involved in pathological conditions such as neurological, immunological and hormonal disorders. A large number of GPCRs have been identified, but whose ligands are not known, are classified as orphan receptors.
-
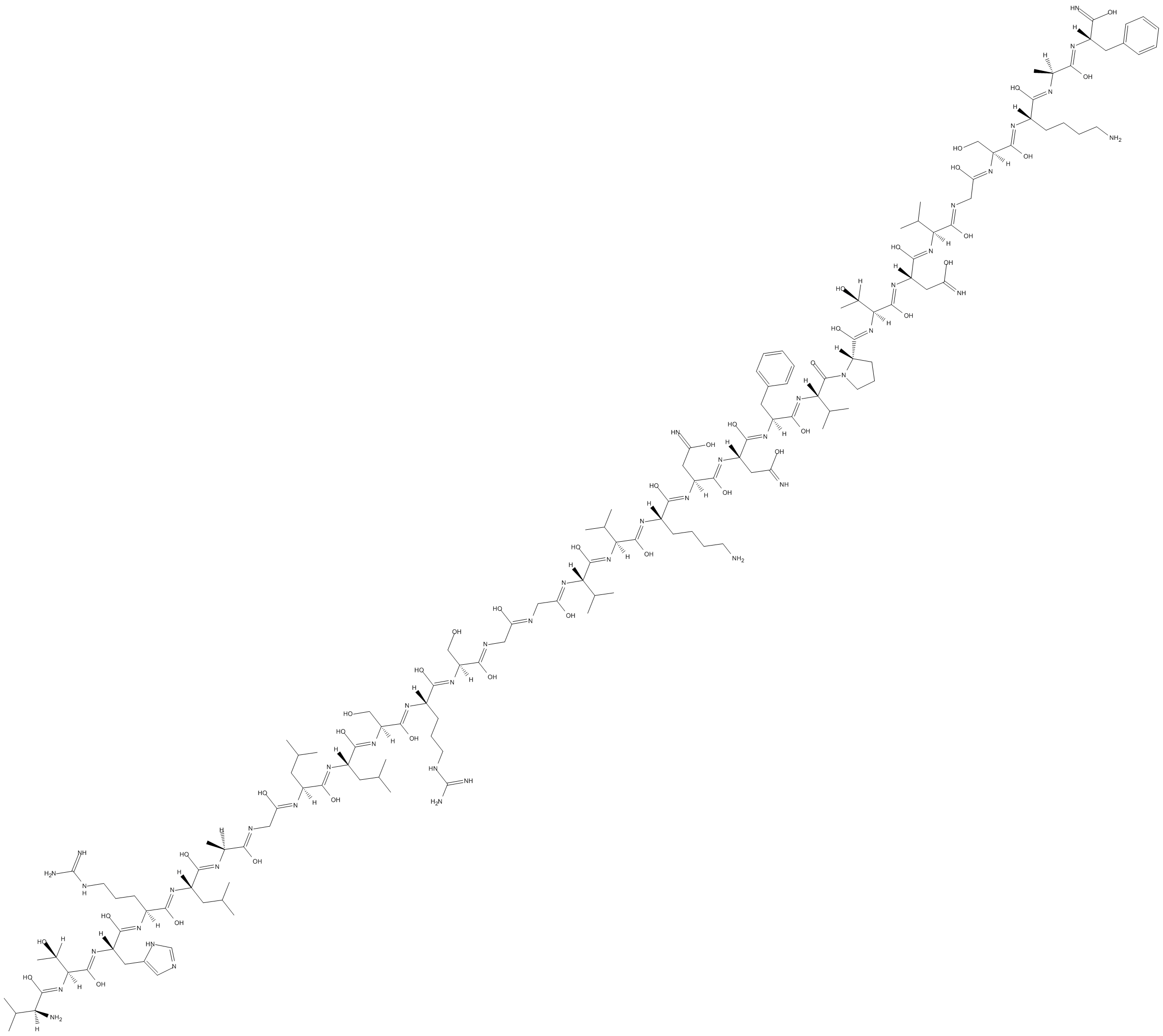 B5073 CGRP 8-37 (human)Summary: Peptide antagonist for CGRP1 receptors
B5073 CGRP 8-37 (human)Summary: Peptide antagonist for CGRP1 receptors -
 B5074 VIP (guinea pig)Summary: Neuropeptide with many biological actions
B5074 VIP (guinea pig)Summary: Neuropeptide with many biological actions -
 B5075 PACAP 1-27Summary: endogenous neuropeptide, stimulator of adenylate cyclase
B5075 PACAP 1-27Summary: endogenous neuropeptide, stimulator of adenylate cyclase -
 B5076 α-helical CRF 9-41Summary: Antagonist of corticotropin releasing factor receptor
B5076 α-helical CRF 9-41Summary: Antagonist of corticotropin releasing factor receptor -
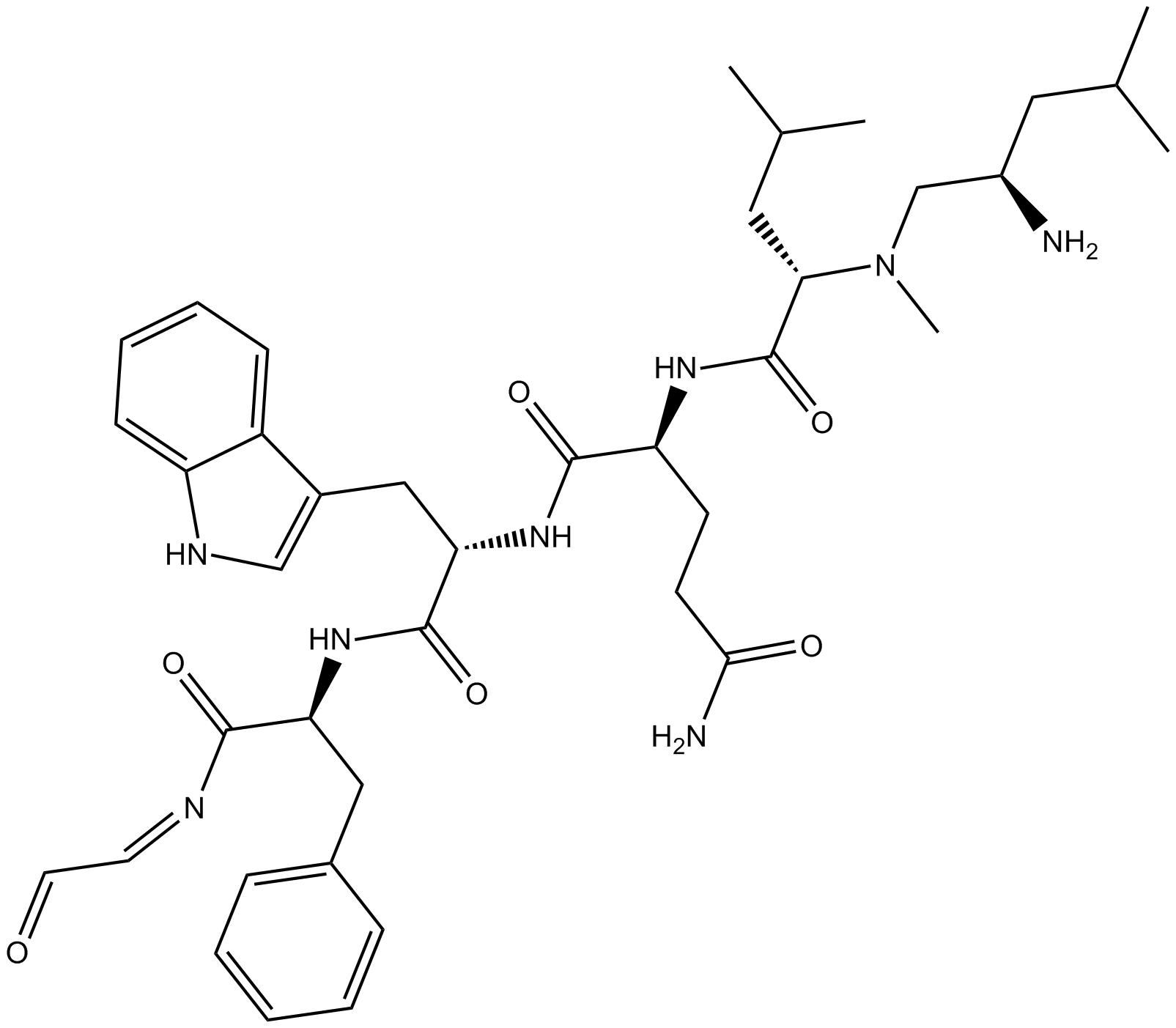 B5078 MDL 29,913Summary: NK2 tachykinin receptor selective antagonist
B5078 MDL 29,913Summary: NK2 tachykinin receptor selective antagonist -
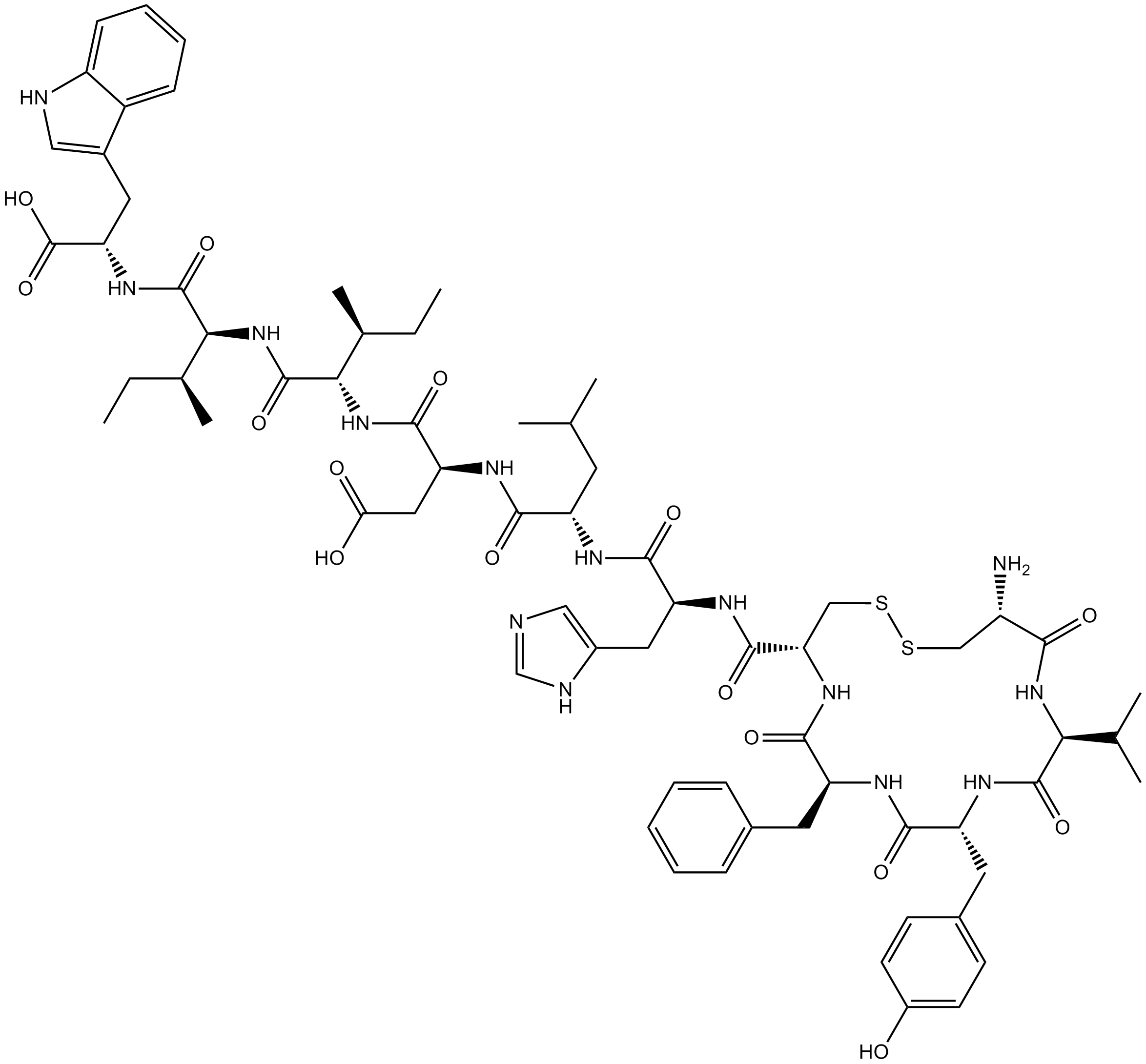 B5079 IRL-1038Summary: ETB endothelin receptor antagonist
B5079 IRL-1038Summary: ETB endothelin receptor antagonist -
![[Ala1,3,11,15]-Endothelin](/pub/media/prod_images/b/5/b5080.png) B5080 [Ala1,3,11,15]-EndothelinSummary: Selective ETB endothelin receptor agonist
B5080 [Ala1,3,11,15]-EndothelinSummary: Selective ETB endothelin receptor agonist -
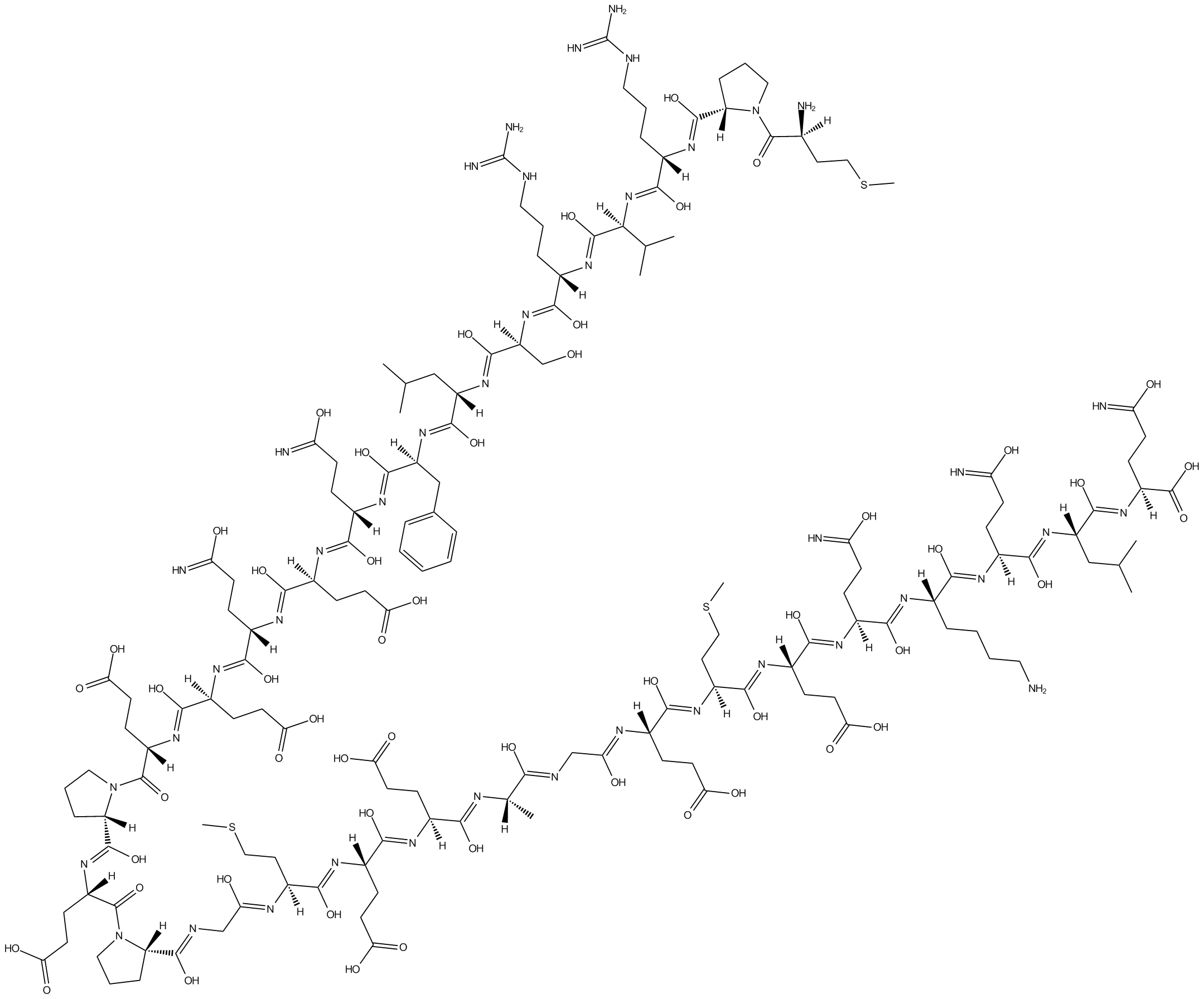 B5081 Nocistatin (human)Summary: Blocker of nociceptin-induced allodynia and hyperalgesia
B5081 Nocistatin (human)Summary: Blocker of nociceptin-induced allodynia and hyperalgesia -
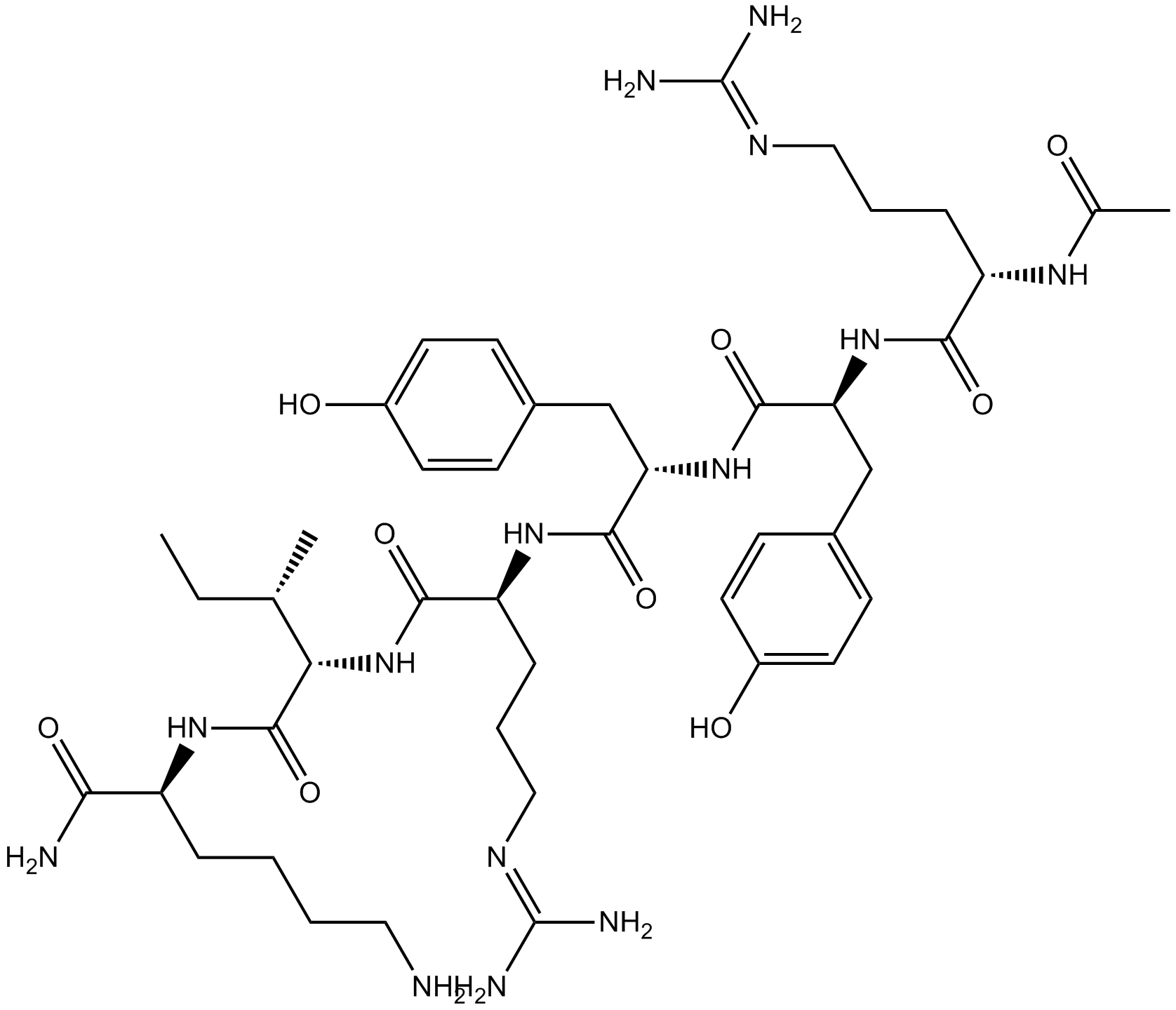 B5085 Ac-RYYRIK-NH2Summary: High affinity ligand for the NOP site
B5085 Ac-RYYRIK-NH2Summary: High affinity ligand for the NOP site -
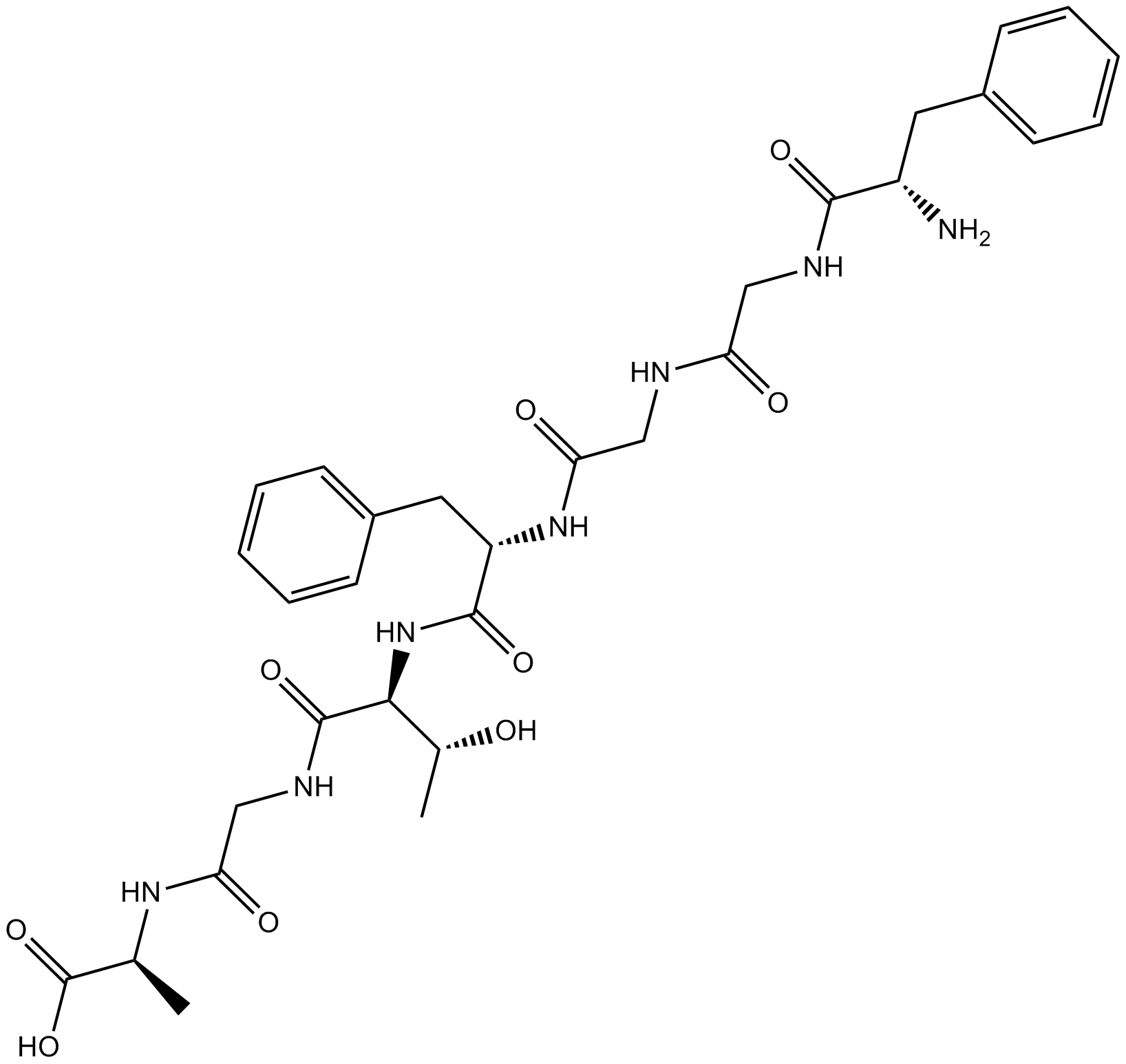 B5087 Nociceptin (1-7)Summary: Bioactive metabolite of nociceptin, antagonist of nociceptin-induced hyperalgesia
B5087 Nociceptin (1-7)Summary: Bioactive metabolite of nociceptin, antagonist of nociceptin-induced hyperalgesia


