Search results for: 'p 22077'
-
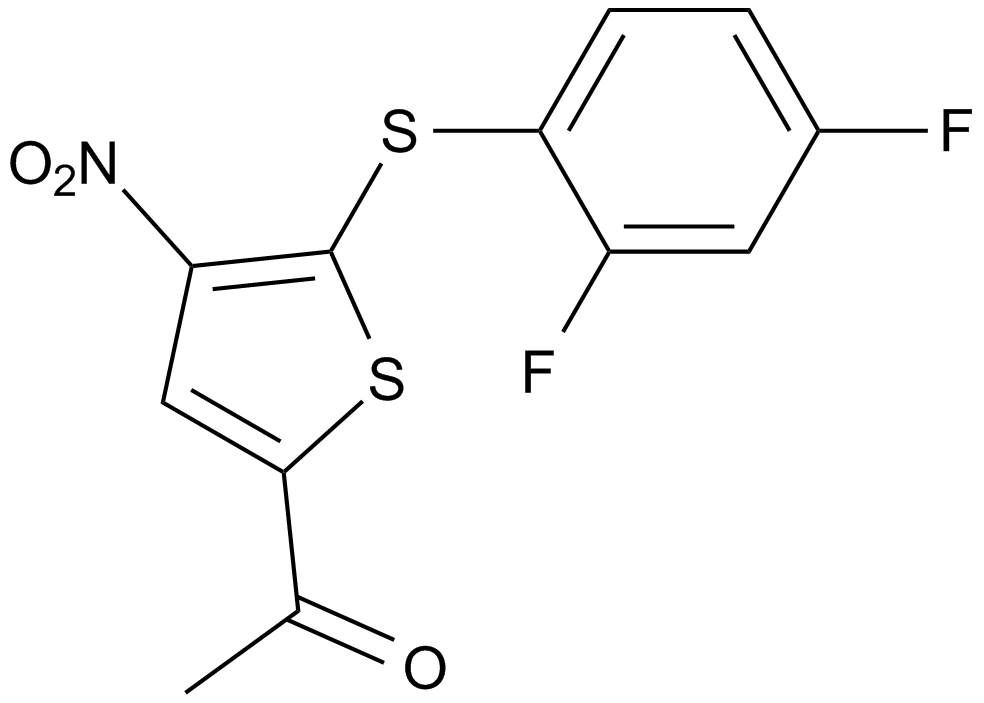 A8198 P 22077Summary: USP7/(DUB)USP47 inhibitor
A8198 P 22077Summary: USP7/(DUB)USP47 inhibitor -
 B6620 Substance PSummary: Sensory neuropeptide and inflammatory mediator
B6620 Substance PSummary: Sensory neuropeptide and inflammatory mediator -
![[Sar9,Met(O2)11]-Substance P](/pub/media/prod_images/b/6/b6623.png) B6623 [Sar9,Met(O2)11]-Substance PSummary: NK1 tachykinin receptor agonist
B6623 [Sar9,Met(O2)11]-Substance PSummary: NK1 tachykinin receptor agonist -
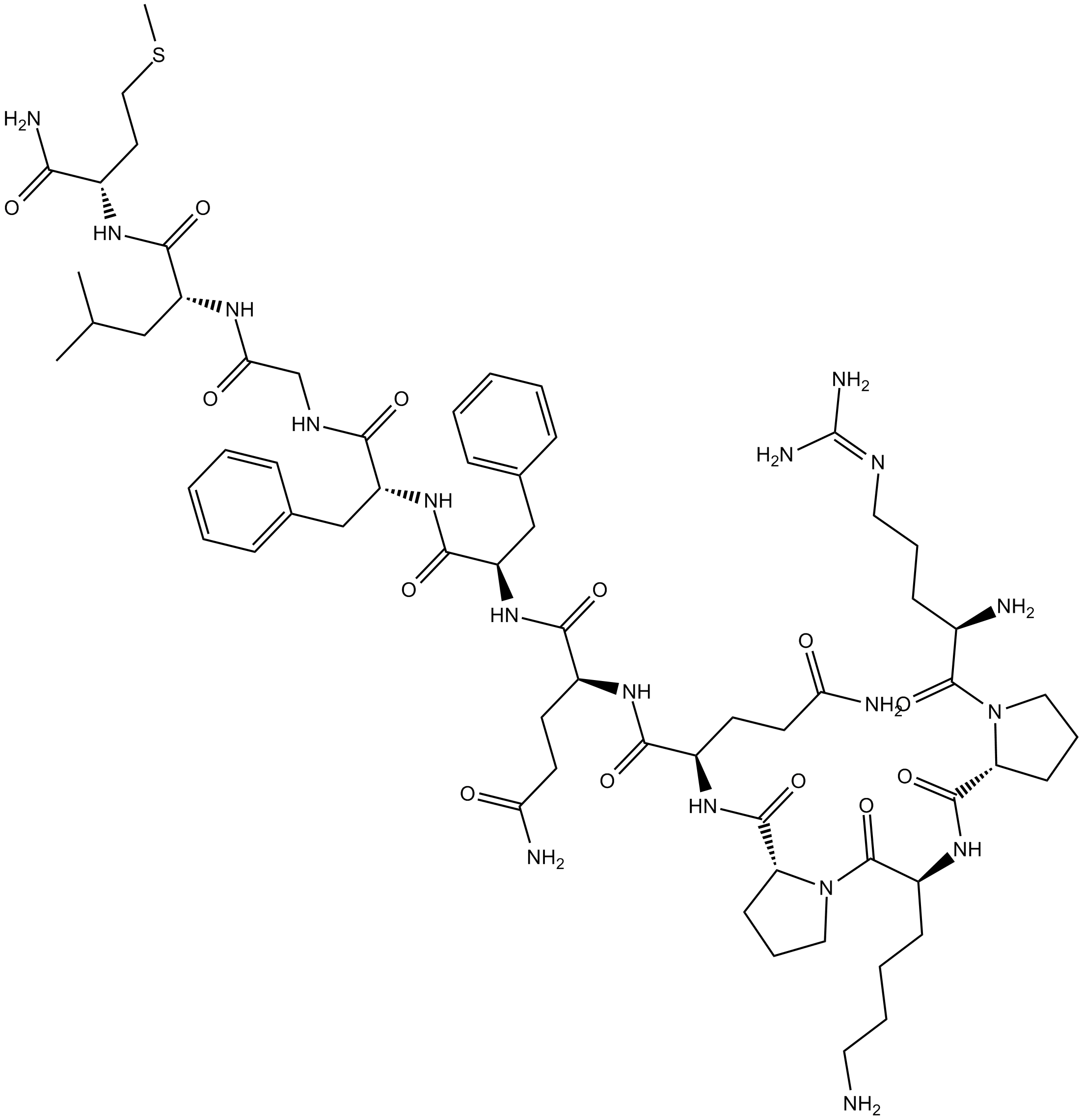
![[D-Trp7,9,10]-Substance P](/pub/media/prod_images/b/6/b6885.png) B6885 [D-Trp7,9,10]-Substance PSummary: inhibits activation of Gq/11 by M1 muscarinic ACh receptors
B6885 [D-Trp7,9,10]-Substance PSummary: inhibits activation of Gq/11 by M1 muscarinic ACh receptors -
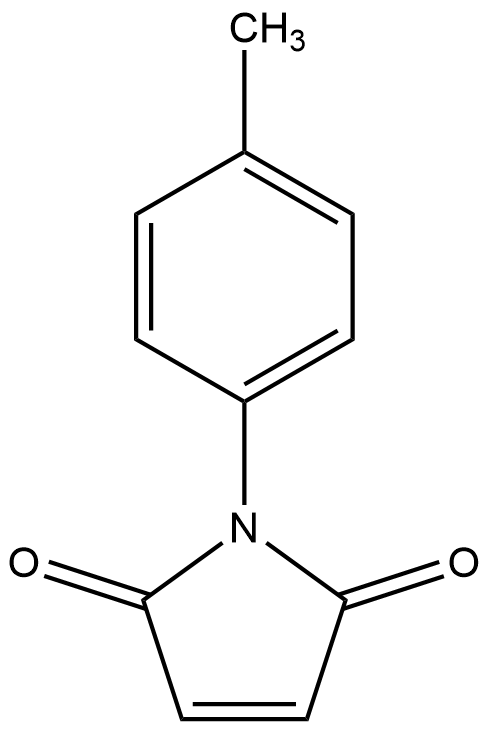 A8995 p-Tolylmaleimide
A8995 p-Tolylmaleimide -
 BA5614 SeptideSummary: Septide ((Pyr6,Pro9)-SubstanceP) is a potent agonist.
BA5614 SeptideSummary: Septide ((Pyr6,Pro9)-SubstanceP) is a potent agonist. -
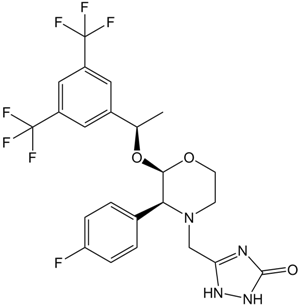 A1684 AprepitantTarget: Substance P/NK1 ReceptorSummary: Substance P (SP) inhibitor
A1684 AprepitantTarget: Substance P/NK1 ReceptorSummary: Substance P (SP) inhibitor -
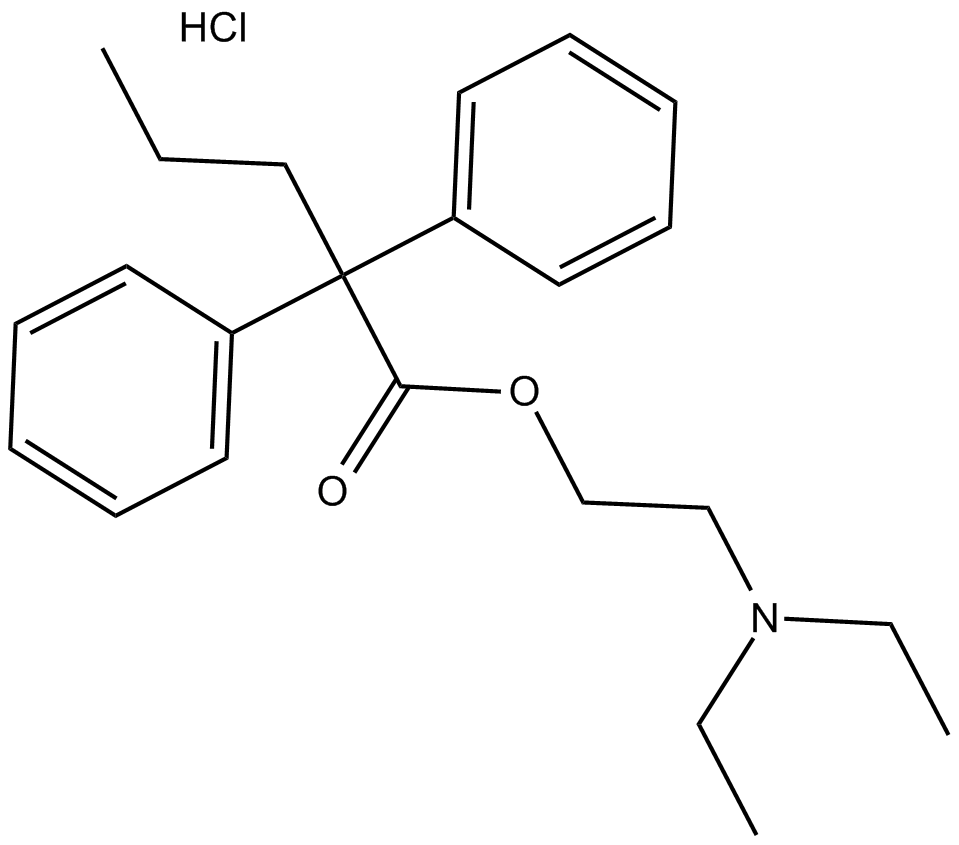 C3560 SKF 525A (hydrochloride)Summary: cytochrome P (CYP)450 inhibitor
C3560 SKF 525A (hydrochloride)Summary: cytochrome P (CYP)450 inhibitor -
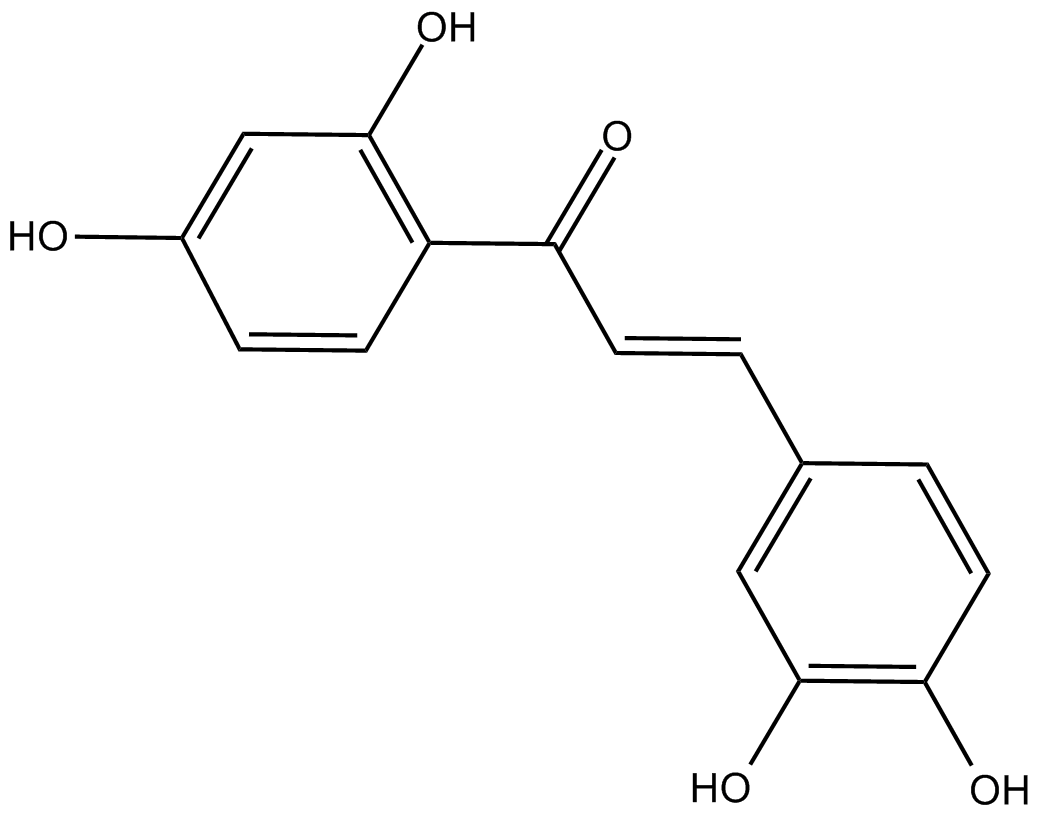 A2689 ButeinTarget: Src|STAT|EGFR|Angiotensin-Converting Enzymes (ACEs)|5-Lipoxygenases|PTK|enoyl-acyl-carrier protein reductaseSummary: Protein kinase inhibitor
A2689 ButeinTarget: Src|STAT|EGFR|Angiotensin-Converting Enzymes (ACEs)|5-Lipoxygenases|PTK|enoyl-acyl-carrier protein reductaseSummary: Protein kinase inhibitor -
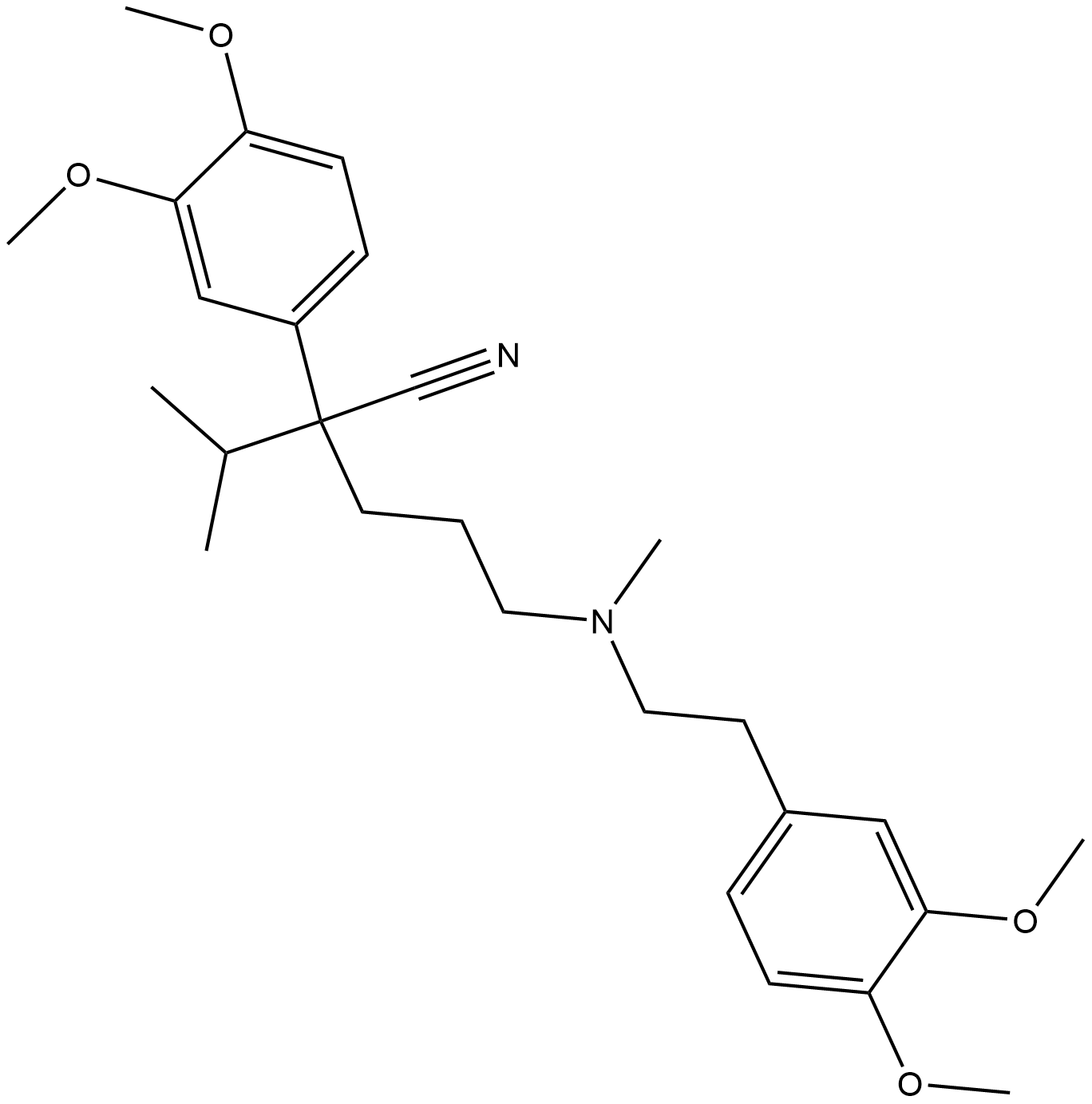 BA6564 VerapamilSummary: Verapamil ((±)-Verapamil) is a calcium channel blocker and a potent orally active first-generation P glycoprotein inhibitor.
BA6564 VerapamilSummary: Verapamil ((±)-Verapamil) is a calcium channel blocker and a potent orally active first-generation P glycoprotein inhibitor.


