Gemcitabine
Gemcitabine is an inhibitor of DNA synthesis [1].
DNA synthesis is a natural creation of deoxyribonucleic acid (DNA) molecules and plays an important role in cell growth.
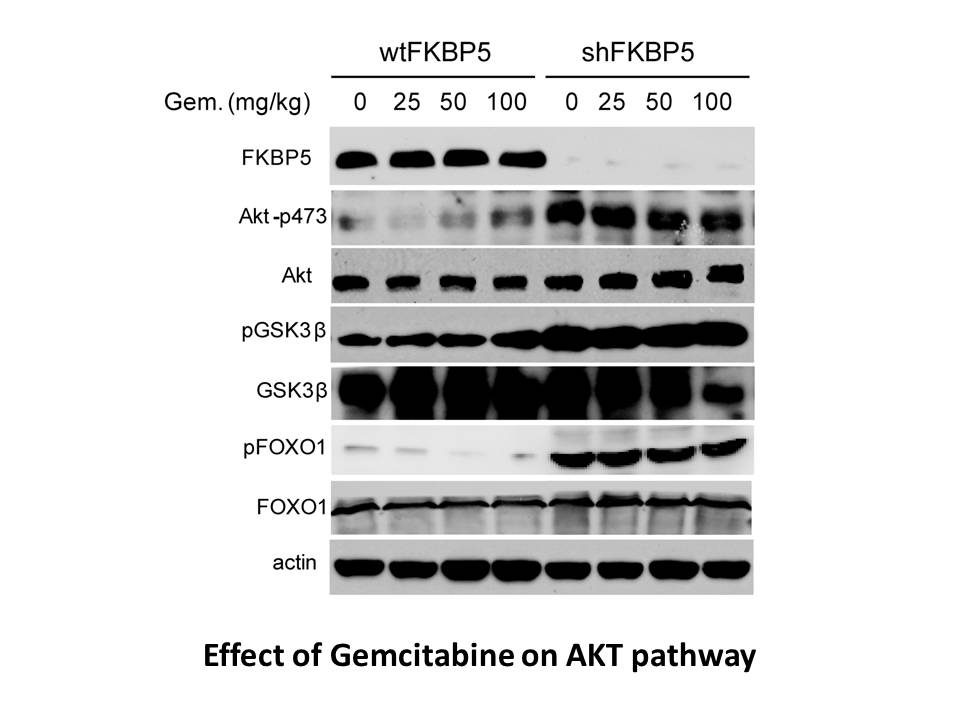
Gemcitabine is an active chemotherapeutic agents that disrupt DNA replication. In tumor cells, gemcitabine activated checkpoint kinase 2 (Chk2) and ataxia-telangiectasia mutated kinase (ATM), which regulated apoptosis, DNA repair and cell-cycle arrest. Also, gemcitabine activated the Rad9-Hus1-Rad1 complex and the protein kinases ATM and ATR and checkpoint kinase 1 (Chk1), which blocked cell-cycle progression and influence DNA repair [1]. Gemcitabine is a DNA synthesis inhibitor with anti-tumor activity. In human osteosarcoma cell lines HOS and MG63, gemcitabine inhibited DNA synthesis and induced apoptosis [2].
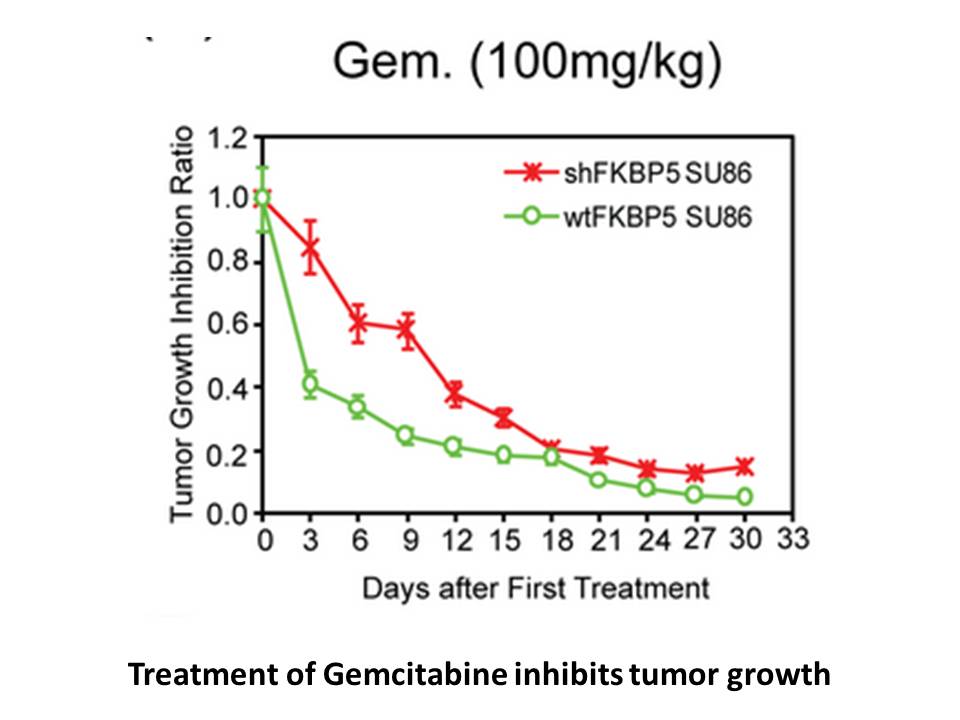
In C3H mice inoculated with murine osteosarcoma cell line LM8, gemcitabine induced cell apoptotics and reduced the size of primary tumor. Also, it inhibited metastatic lesions in the lung [2]. In C57Bl/6 mice infected with LP-BM5 murine leukemia virus, gemcitabine significantly inhibited disease progression. Also, gemcitabine reduced spleen size, provirus levels and plasma IgM [3].
References:
[1] Karnitz LM, Flatten KS, Wagner JM, et al. Gemcitabine-induced activation of checkpoint signaling pathways that affect tumor cell survival. Mol Pharmacol, 2005, 68 (6): 1636-1644.
[2] Ando T, Ichikawa J, Okamoto A, et al. Gemcitabine inhibits viability, growth, and metastasis of osteosarcoma cell lines. J Orthop Res, 2005, 23 (4): 964-969.
[3] Clouser CL, Holtz CM, Mullett M, et al. Analysis of the ex vivo and in vivo antiretroviral activity of gemcitabine. PLoS One, 2011, 6 (1): e15840.
- 1. Ning Zhang, Linmao Sun, et al. "Cholangiocarcinoma PDHA1 succinylation suppresses macrophage antigen presentation via alpha-ketoglutaric acid accumulation." Nat Commun. 2025 Apr 3;16(1):3177 PMID: 40180922
- 2. Jian Jiang, Tianci Shen, et al. "FOXM1, a super enhancer-associated gene, is related to poorer prognosis and gemcitabine resistance in pancreatic cancer." Cell Biochem Biophys. 2025 Jun;83(2):2441-2452 PMID: 39899193
- 3. Ling He, Xiongbing Li, Yaolan Zhen. "METTL3/IGF2BP3 axis promotes gemcitabine resistance of pancreatic cancer cells through regulating USP33-mediated PAK1 deubiquitination and degradation." Naunyn Schmiedebergs Arch Pharmacol. 2025 Oct 18 PMID: 41108357
- 4. Wenyan Shi, Zi Ye, et al. "Rapid Separation and Detection of Drugs in Complex Biological Matrix Using TD-CDI Mass Spectrometer." Biosensors (Basel). 2024 May 25;14(6):271 PMID: 38920575
- 5. SJ Cutty, FA Hughes, et al. "Pro-survival roles for p21(Cip1/Waf1) in Non-Small Cell Lung Cancer." bioRxiv. May 21, 2024
- 6. Alain Valdivia, Adebimpe Adefolaju, et al. "A Novel ex-vivo platform for personalized treatment in metastatic ovarian cancer." bioRxiv. March 16, 2024
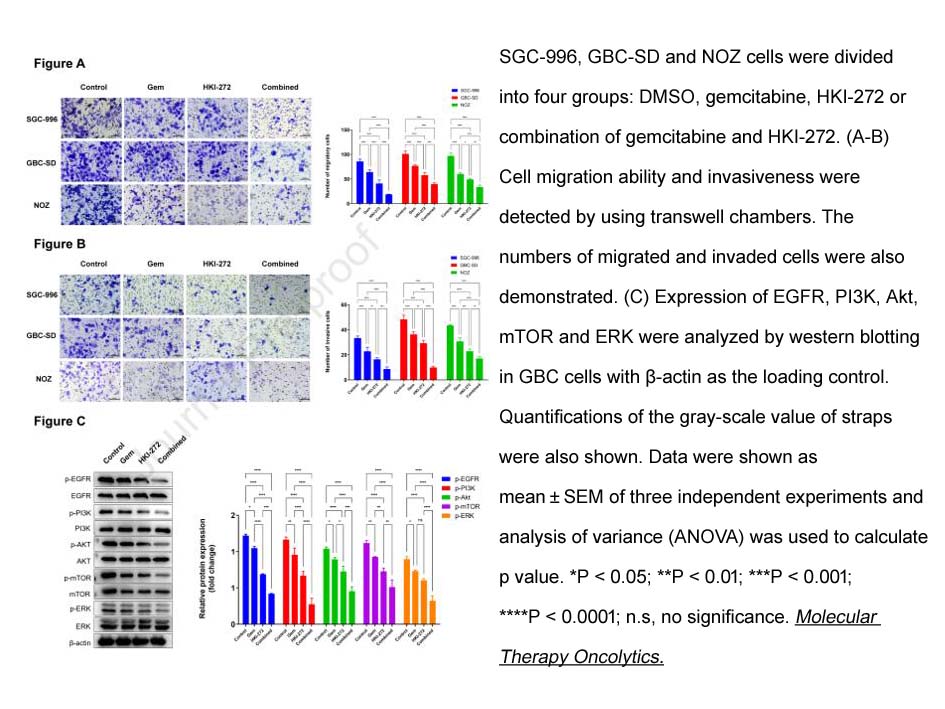
- 7. Xuli Yang, Tao Chen, et al. "HKI-272 contributes to gemcitabine-mediated anti-proliferative and anti-metastatic effects through EGFR suppression in gallbladder cancer." Mol Ther Oncolytics. 2022 Oct 8;27:126-140 PMID: 36321135
- 8. Xuejiao Wu, Jia Huang, et al. "MicroRNA-221-3p is related to survival and promotes tumour progression in pancreatic cancer: a comprehensive study on functions and clinicopathological value." Cancer Cell Int 2020 Sep 10;20:443 PMID: 32943991
- 9. Lin KH, Xie A, et al. "Systematic Dissection of the Metabolic-Apoptotic Interface in AML Reveals Heme Biosynthesis to Be a Regulator of Drug Sensitivity." Cell Metab. 2019 Feb 5. pii: S1550-4131(19)30011-7 PMID: 30773463
| Physical Appearance | A solid |
| Storage | Store at -20°C |
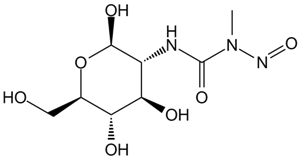
| M.Wt | 263.2 |
| Cas No. | 95058-81-4 |
| Formula | C9H11F2N3O4 |
| Solubility | ≥11.75 mg/mL in H2O with gentle warming; ≥26.34 mg/mL in DMSO; ≥7.54 mg/mL in EtOH with ultrasonic |
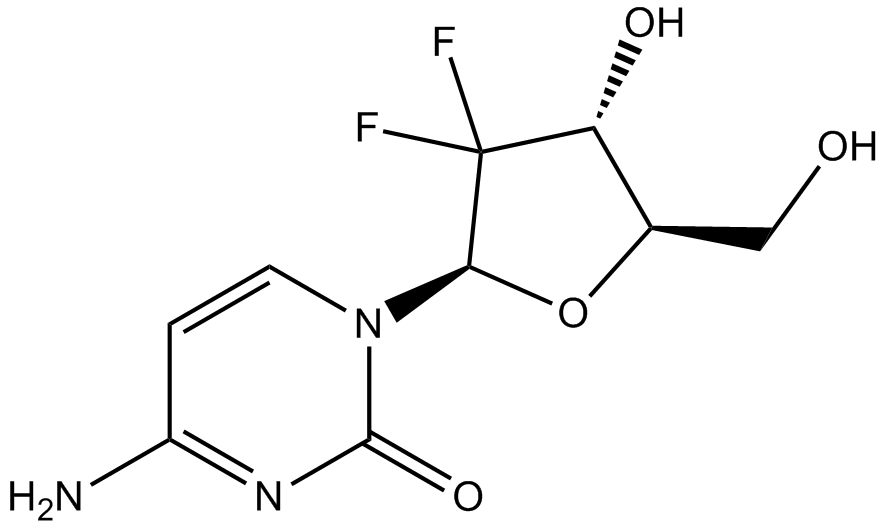
| Chemical Name | 4-amino-1-[(2R,4R,5R)-3,3-difluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidin-2-one |
| SDF | Download SDF |
| Canonical SMILES | NC(C=CN1[C@@H](C2(F)F)O[C@H](CO)[C@H]2O)=NC1=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
HeLa cells, K562 cells, HOS and MG63 cell lines. |
|
Preparation method |
The solubility of this compound in DMSO >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37°C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20°C for several months. |
|
Reacting condition |
100 nM gemcitabine for 3 h in HeLa cells for immunofluorescence, 500 nM gemcitabine for 6 h for SDS-PAGE |
|
Applications |
In Hela cells and K562 cells, gemcitabine activated both the ATR/Chk1 and ATM/Chk2 signaling pathways. (ATR: ataxia-telangiectasia mutated and Rad3-related kinase; Chk: checkpoint kinase; ATM: ataxia-telangiectasia mutated kinase). Gemcitabine is a DNA synthesis inhibitor with anti-tumor activity. In human osteosarcoma cell lines HOS and MG63, gemcitabine inhibited DNA synthesis and induced apoptosis. |
| Animal experiment [2]: | |
|
Animal models |
Female C57BL/6 mice infected with LP-BM5 MuLV |
|
Dosage form |
1, 2, 4 mg/kg/day for 8 week by injection. |
|
Application |
Mice treated with 1 or 2 mg/kg/day had an average ratio of spleen to body weight that was significantly lower than the infected with virus, untreated mice. Treatment with gemcitabine decreased MAIDS associated lesions in the lymph nodes. IgM levels from mice treated with 2 mg/kg/day of gemcitabine were significantly lower than that seen in the uninfected animals. Gemcitabine decreased provirus levels. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Karnitz LM, Flatten KS, Wagner JM, et al. Gemcitabine-induced activation of checkpoint signaling pathways that affect tumor cell survival. Mol Pharmacol, 2005, 68 (6): 1636-1644. [2] Ando T, Ichikawa J, Okamoto A, et al. Gemcitabine inhibits viability, growth, and metastasis of osteosarcoma cell lines. J Orthop Res, 2005, 23 (4): 964-969. [3] Clouser CL, Holtz CM, Mullett M, et al. Analysis of the ex vivo and in vivo antiretroviral activity of gemcitabine. PLoS One, 2011, 6 (1): e15840. |
|
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
Related Biological Data
Related Biological Data
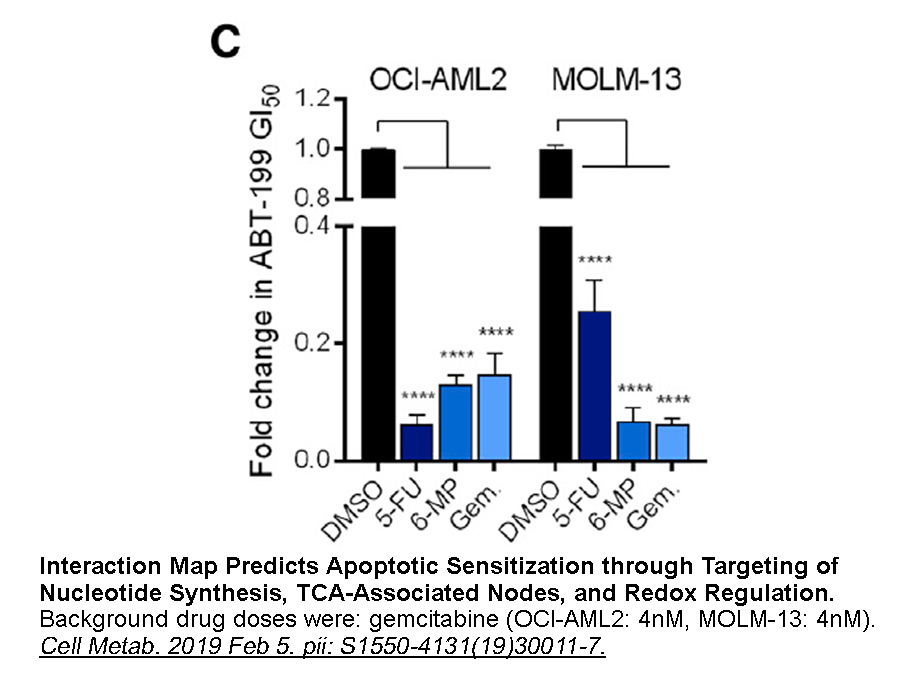
Related Biological Data
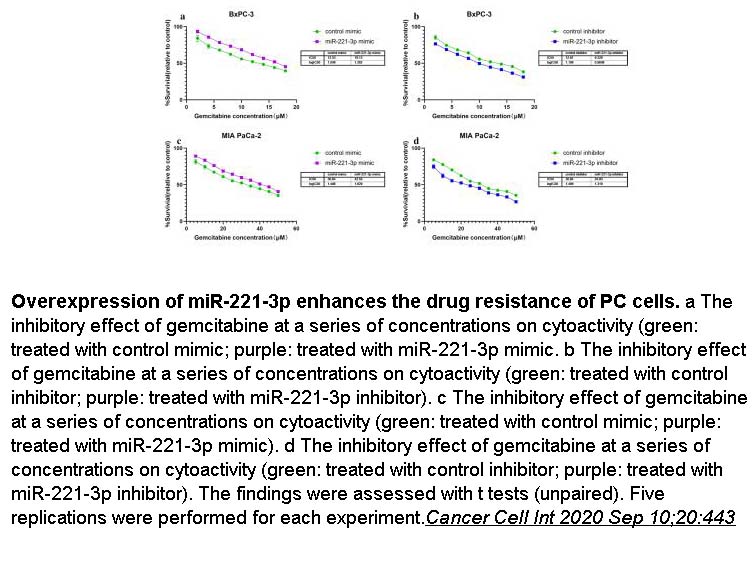
Related Biological Data