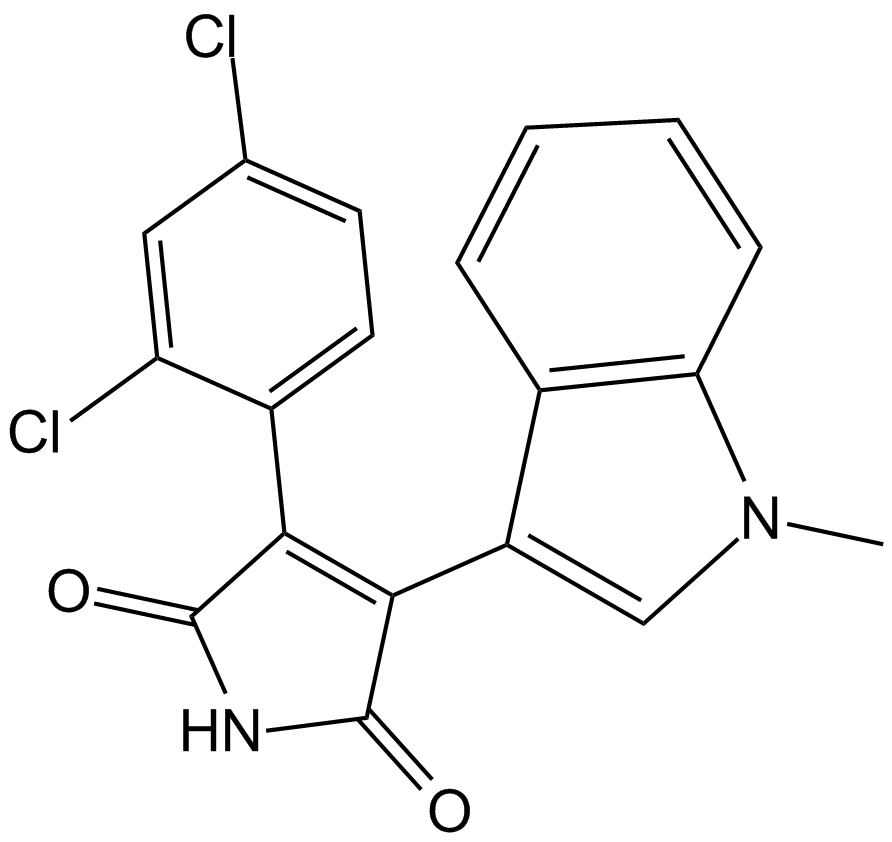
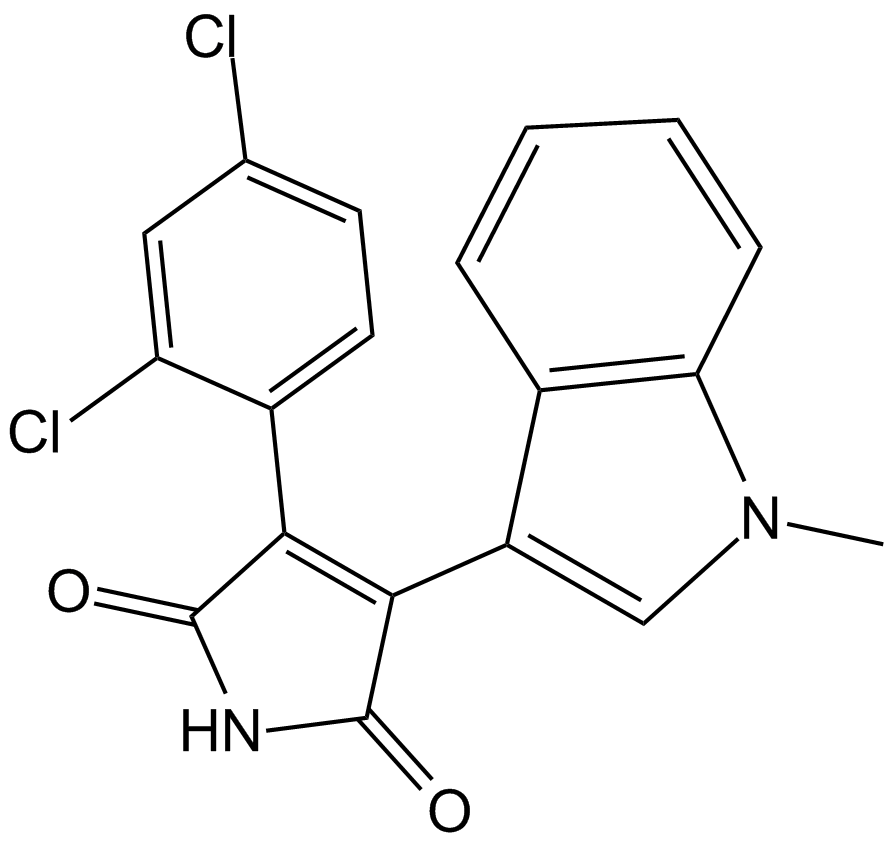
SB 216763
SB-216763 is a Potent and selective inhibitor of glycogen synthase kinase-3 (GSK-3) with IC50 of 34.3 nM. It competes with ATP and potently inhibits the activity against both GSK-3α and GSK-3β.
GSK-3, one of several known substrates of PKB, is a serine/threonine protein kinase that mediates the addition of phosphate molecules onto serine and threonine amino acid residues.
SB-216763 treatment can protect neurones of both central and peripheral nervous system from cell death which is mediated by PI3K pathway ex vivo [1]. This component was also shown to maintain the pluripotency of mouse embryonic stem cells (mESCs) without exogenous leukemia inhibitory factor (LIF) when cultured on mouse embryonic fibroblasts (MEFs) [2].
The component has also been used extensively in various animal models to study the role of GSK-3. In a bleomycin-induced mice lung fibrosis model, SB-216763 administration protected mice from lung inflammation and the subsequent fibrosis, which was associated with decreased production of inflammatory cytokine by macrophages [3]. Besides, treatment of SB-216763 reduced infarct size and prevented cardiac ischemia in a model of regional myocardial ischemia-reperfusion in rats [4].
References:
1. Cross DA, Culbert AA, Chalmers KA, Facci L, Skaper SD, Reith AD. Selective small-molecule inhibitors of glycogen synthase kinase-3 activity protect primary neurones from death. J Neurochem 2001,77:94-102.
2. Kirby LA, Schott JT, Noble BL, Mendez DC, Caseley PS, Peterson SC, et al. Glycogen synthase kinase 3 (GSK3) inhibitor, SB-216763, promotes pluripotency in mouse embryonic stem cells. PLoS One 2012,7:e39329.
3. Gurrieri C, Piazza F, Gnoato M, Montini B, Biasutto L, Gattazzo C, et al. 3-(2,4-dichlorophenyl)-4-(1-methyl-1H-indol-3-yl)-1H-pyrrole-2,5-dione (SB216763), a glycogen synthase kinase-3 inhibitor, displays therapeutic properties in a mouse model of pulmonary inflammation and fibrosis. J Pharmacol Exp Ther 2010,332:785-794.
4. Obame FN, Plin-Mercier C, Assaly R, Zini R, Dubois-Rande JL, Berdeaux A, et al. Cardioprotective effect of morphine and a blocker of glycogen synthase kinase 3 beta, SB216763 [3-(2,4-dichlorophenyl)-4(1-methyl-1H-indol-3-yl)-1H-pyrrole-2,5-dione], via inhibition of the mitochondrial permeability transition pore. J Pharmacol Exp Ther 2008,326:252-258.
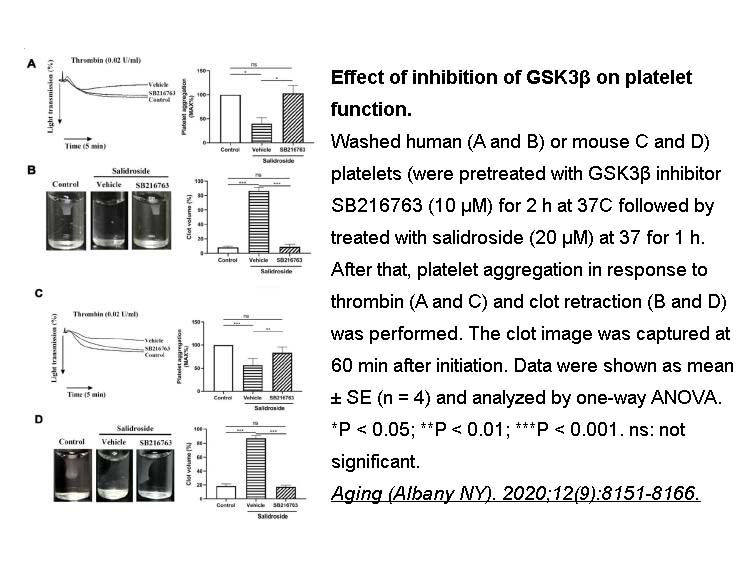
- 1. Wei G, Xu X, et al. "Salidroside inhibits platelet function and thrombus formation through AKT/GSK3β signaling pathway." Aging (Albany NY). 2020;12(9):8151-8166. PMID:32352928
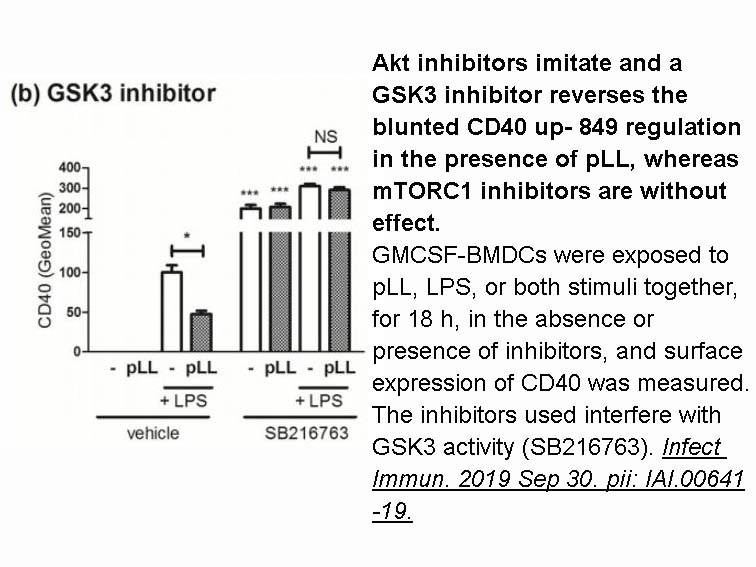
- 2. Pittini á, Martínez-Acosta YE, et al. "Particles from the Echinococcus granulosus laminated layer inhibit CD40 upregulation in dendritic cells by interfering with Akt activation." Infect Immun. 2019 Sep 30. pii: IAI.00641-19. PMID:31570562
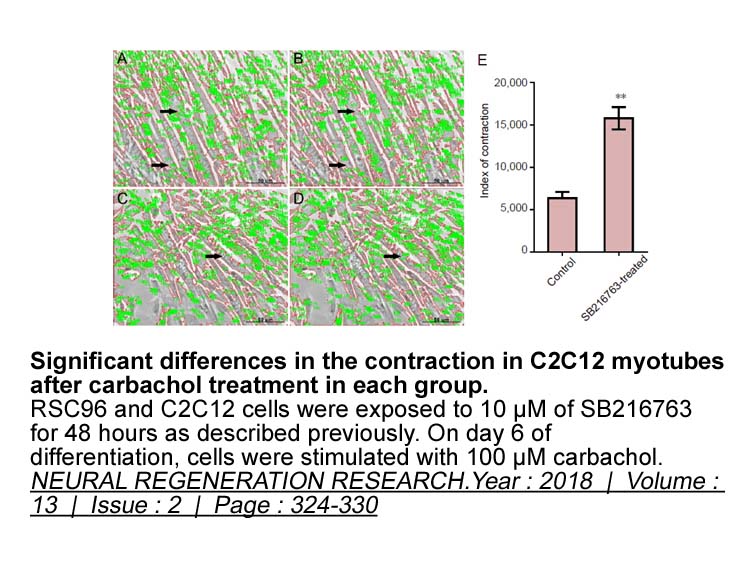
- 3. Weng J, Wang YH, et al. "GSK3β inhibitor promotes myelination and mitigates muscle atrophy after peripheral nerve injury." Neural Regen Res. 2018 Feb;13(2):324-330. PMID:29557384
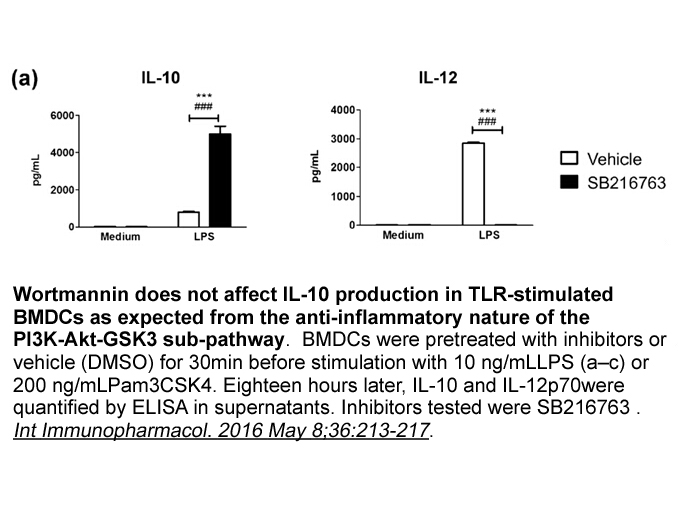
- 4. Pittini á, Casaravilla C, et al. "Pharmacological inhibition of PI3K class III enhances the production of pro- and anti-inflammatory cytokines in dendritic cells stimulated by TLR agonists." Int Immunopharmacol. 2016 May 8;36:213-217. PMID:27168056
- 5. Dr.Alvaro Diaz. "Condicionamiento de células dendríticas por la capa laminar de Echinococcus granulosus: búsqueda de agonistas y mecanismos a nivel de se?alizacón." colibri.udelar.edu.uy.2016.
| Storage | Store at RT |
| M.Wt | 371.22 |
| Cas No. | 280744-09-4 |
| Formula | C19H12Cl2N2O2 |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥56.8 mg/mL in DMSO |
| Chemical Name | 3-(2,4-dichlorophenyl)-4-(1-methylindol-3-yl)pyrrole-2,5-dione |
| SDF | Download SDF |
| Canonical SMILES | C[n]1c(cccc2)c2c(C(C(NC2=O)=O)=C2c(ccc(Cl)c2)c2Cl)c1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
GSK-3 activity assay |
GSK-3 kinase activity was measured, in the presence of various concentrations of SB 216763, in a reaction mixture containing final concentrations of 1 nM human GSK-3α, 50 mM MOPS pH 7.0, 0.2 mM EDTA, 10 mM Mg-acetate, 7.5 mM β-mercaptoethanol, 5% (w/v) glycerol, 0.01% (w/v) Tween-20, 10% (v/v) DMSO, and 28 μM GS-2 peptide substrate. The GS-2 peptide sequence corresponds to a region of glycogen synthase that was phosphorylated by GSK-3. The assay was initiated by the addition of 0.34 μCi [33P]γ-ATP. The total ATP concentration was 10 μM. Following 30 mins incubation at room temperature the assay was stopped by the addition of one third assay volume of 2.5% (v/v) H3PO4 containing 21 mM ATP. Samples were spotted onto P30 phosphocellulose mats and washed six times in 0.5% (v/v) H3PO4. The filter mats were sealed into sample bags containing Wallac betaplate scintillation fluid. 33P incorporation into the substrate peptide was determined by counting the mats in a Wallac microbeta scintillation counter. |
| Cell experiment [2]: | |
|
Cell lines |
Cerebellar granule neurons |
|
Preparation method |
The solubility of this compound in DMSO is >56.8mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20 °C for several months. |
|
Reaction Conditions |
0.1 ~ 10 μM; 48 hrs |
|
Applications |
SB 216763 concentration-dependently reduced the death of cerebellar granule neurons induced by potassium-deprivation or LY-294002 treatment. 3 μM SB 216763 exhibited the maximal neuroprotection effect. |
| Animal experiment [3]: | |
|
Animal models |
Bleomycin (BLM)-induced lung inflammation and fibrosis mouse model |
|
Dosage form |
20 mg/kg; i.v. |
|
Applications |
In a Bleomycin (BLM)-induced lung inflammation and fibrosis mouse model, SB 216763 significantly prevented lung inflammation and subsequent fibrosis through significantly blocking the production of inflammatory cytokines MCP-1 and TNF-α by macrophages. Moreover, it significantly improved the survival, and markedly reduced alveolitis by inhibiting alveolar epithelial cell damage. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Coghlan MP, Culbert AA, Cross DA, Corcoran SL, Yates JW, Pearce NJ, Rausch OL, Murphy GJ, Carter PS, Roxbee Cox L, Mills D, Brown MJ, Haigh D, Ward RW, Smith DG, Murray KJ, Reith AD, Holder JC. Selective small molecule inhibitors of glycogen synthase kinase-3 modulate glycogen metabolism and gene transcription. Chem Biol. 2000 Oct;7(10):793-803. [2]. Cross DA, Culbert AA, Chalmers KA, Facci L, Skaper SD, Reith AD. Selective small-molecule inhibitors of glycogen synthase kinase-3 activity protect primary neurones from death. J Neurochem 2001,77:94-102. [3] Gurrieri C, Piazza F, Gnoato M, Montini B, Biasutto L, Gattazzo C, et al. 3-(2,4-dichlorophenyl)-4-(1-methyl-1H-indol-3-yl)-1H-pyrrole-2,5-dione (SB216763), a glycogen synthase kinase-3 inhibitor, displays therapeutic properties in a mouse model of pulmonary inflammation and fibrosis. J Pharmacol Exp Ther 2010,332:785-794. |
|
| Description | SB216763 is a potent and selective inhibitor of GSK-3 with an IC50 value of 34.3 nM. | |||||
| Targets | GSK-3α | GSK-3β | ||||
| IC50 | 34.3 nM | ~34.3 nM | ||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data