Search results for: 'probes dyes cyanine dyes'
-
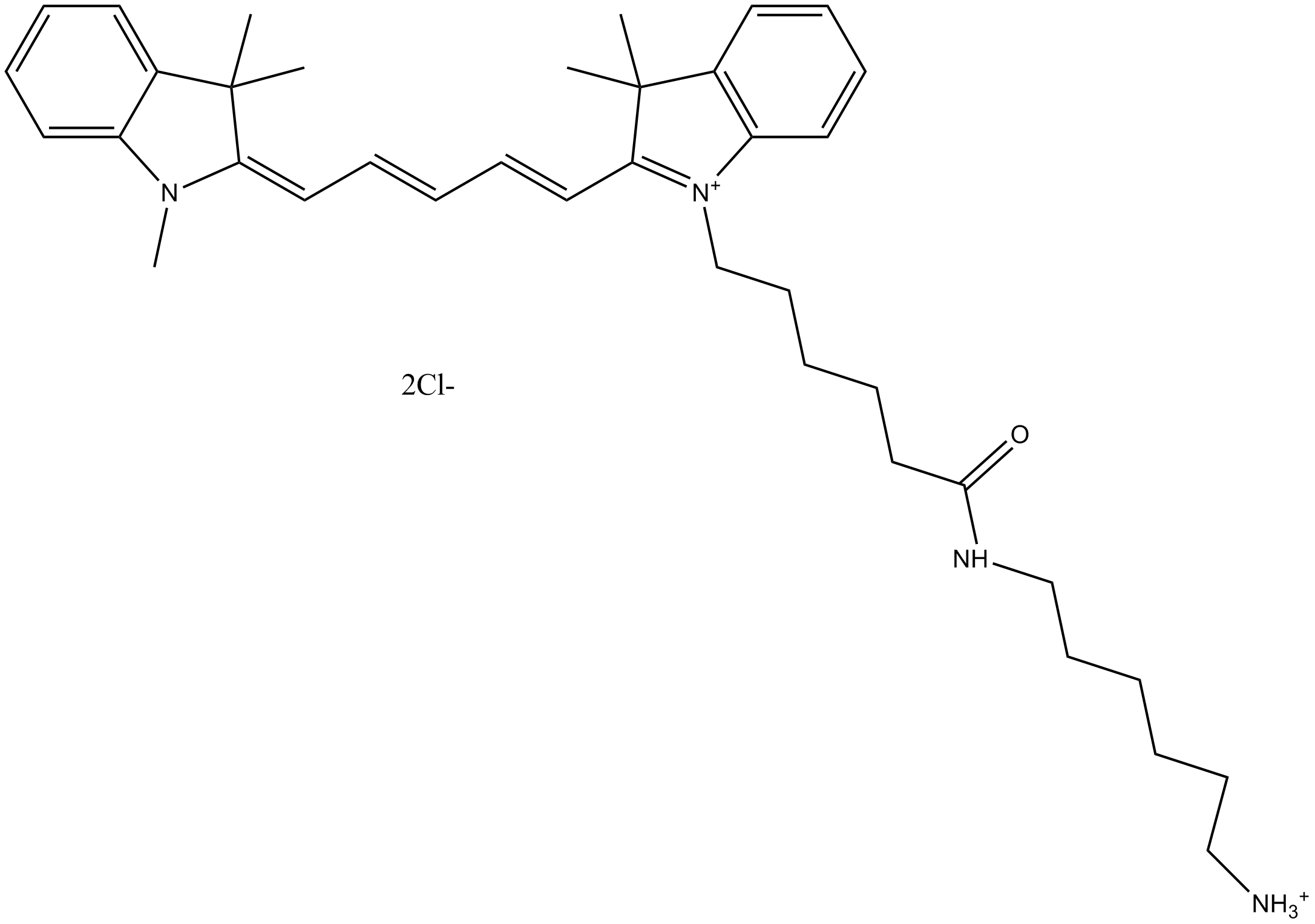 A8143 Cy5 amine (non-sulfonated)1 CitationSummary: can be conjugated with reactive groups such as NHS esters, carboxy groups (after carbodiimide activation), and epoxides.
A8143 Cy5 amine (non-sulfonated)1 CitationSummary: can be conjugated with reactive groups such as NHS esters, carboxy groups (after carbodiimide activation), and epoxides. -
 B8163 Cyanine 3-AA-CTP
B8163 Cyanine 3-AA-CTP -
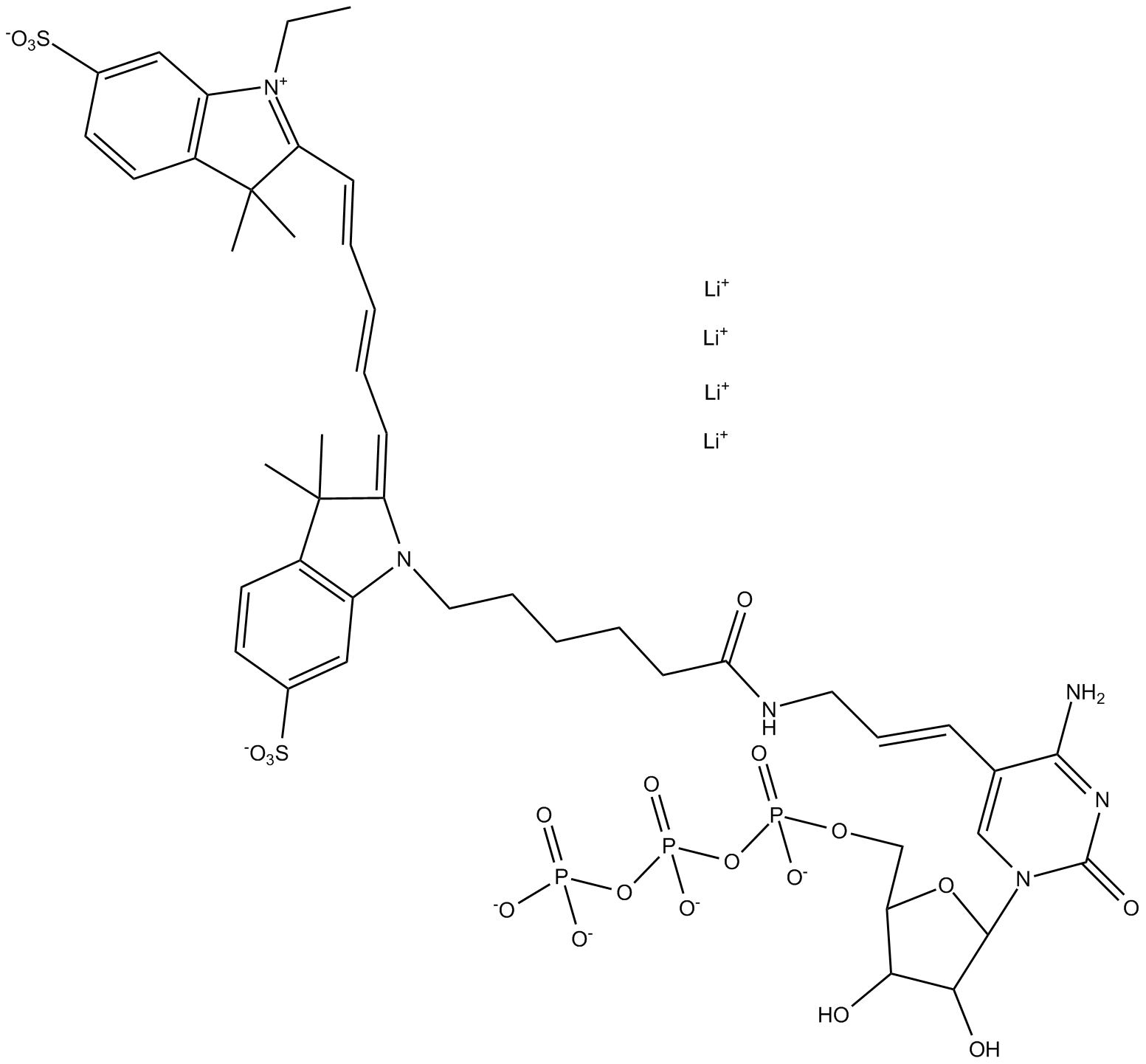 B8165 Cyanine 5-AA-CTPSummary: Cytidine triphosphate modified with Cy5 dye for RNA labeling
B8165 Cyanine 5-AA-CTPSummary: Cytidine triphosphate modified with Cy5 dye for RNA labeling -
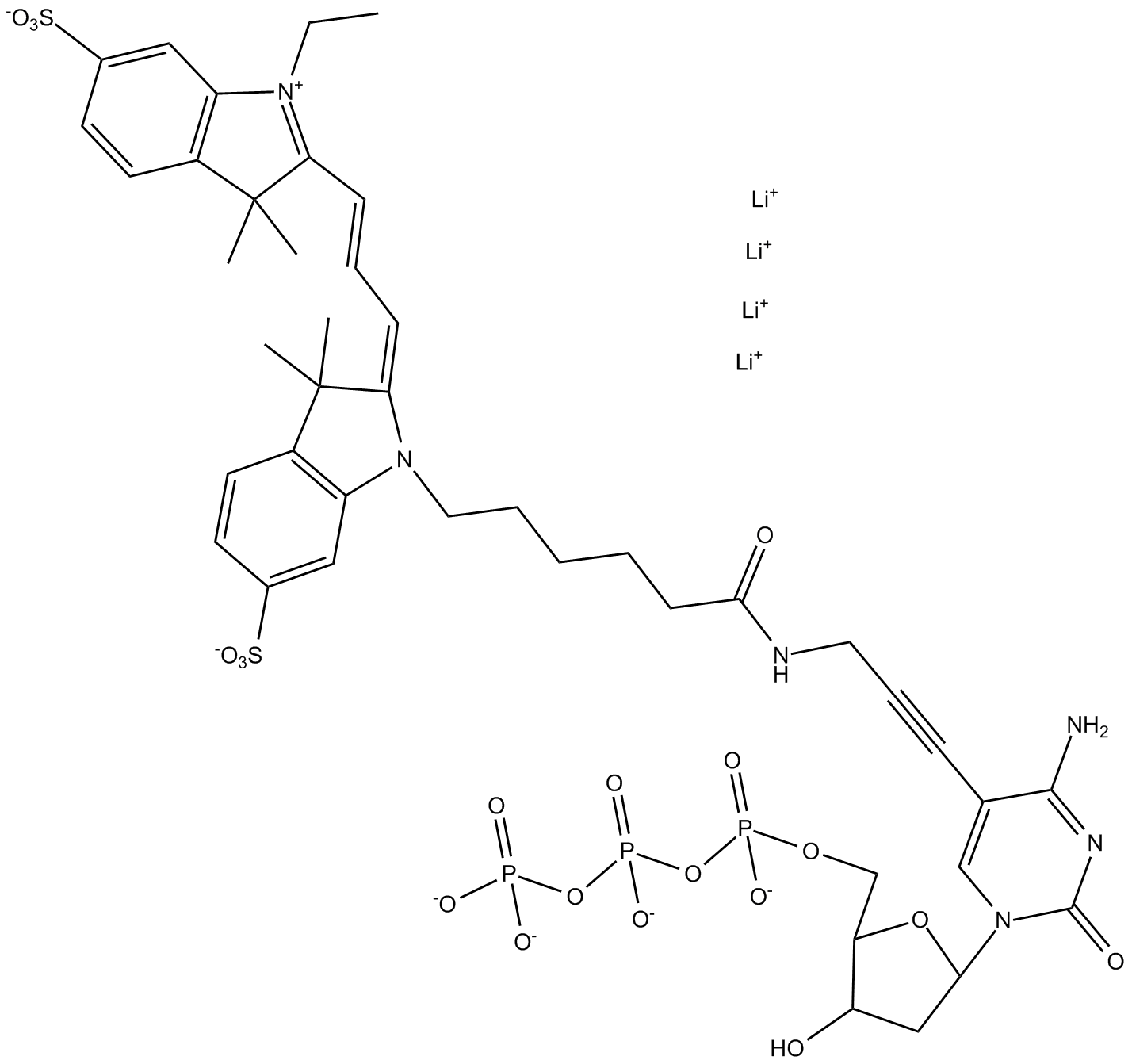 B8159 Cyanine 3-dCTPSummary: Direct enzymatic labeling of DNA/cDNA e.g. by PCR and Nick Translation
B8159 Cyanine 3-dCTPSummary: Direct enzymatic labeling of DNA/cDNA e.g. by PCR and Nick Translation -
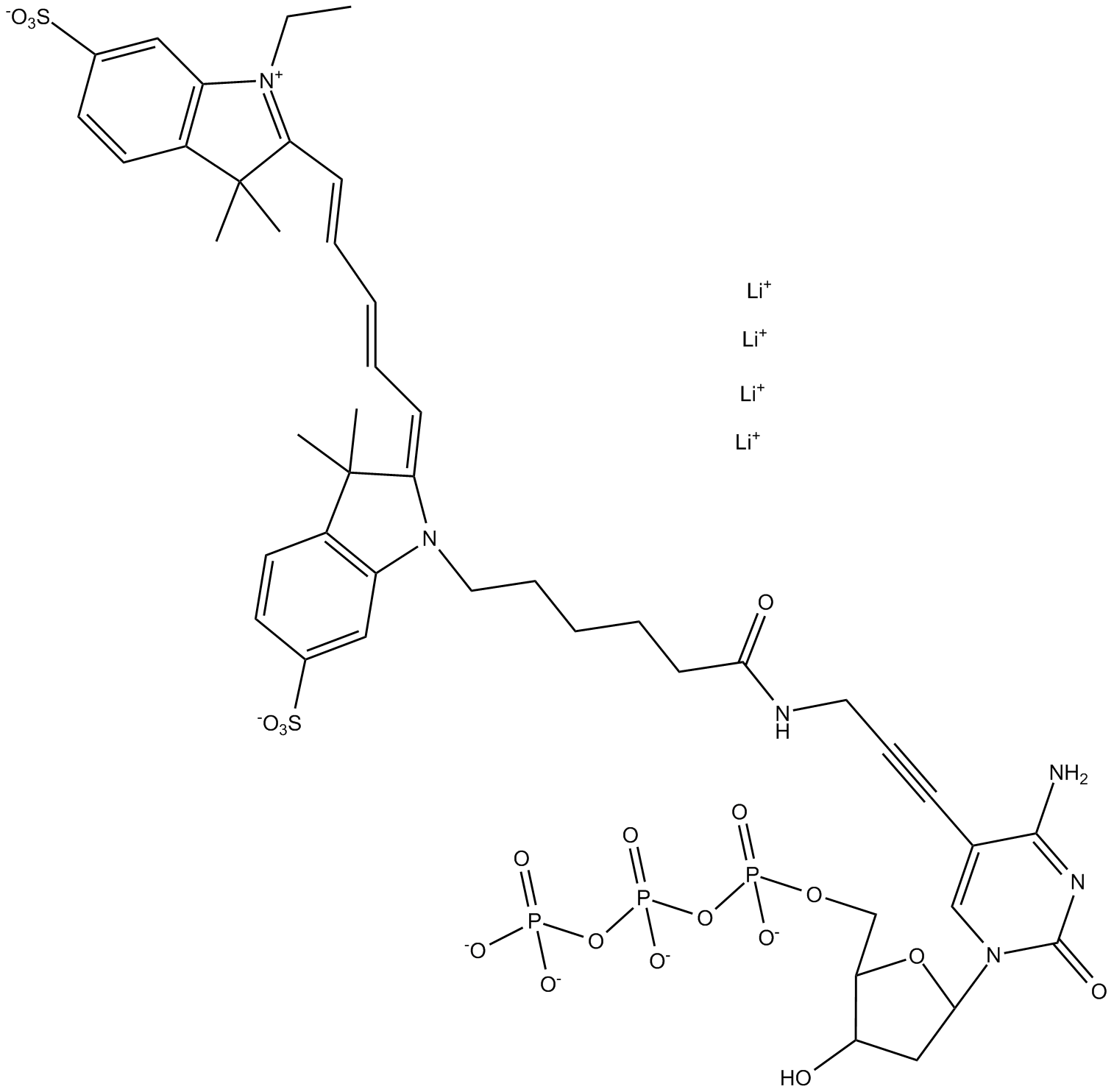 B8161 Cyanine 5-dCTP
B8161 Cyanine 5-dCTP -
 B8162 Cyanine 5-dUTPSummary: A fluorescently-labeled nucleotide analog
B8162 Cyanine 5-dUTPSummary: A fluorescently-labeled nucleotide analog -
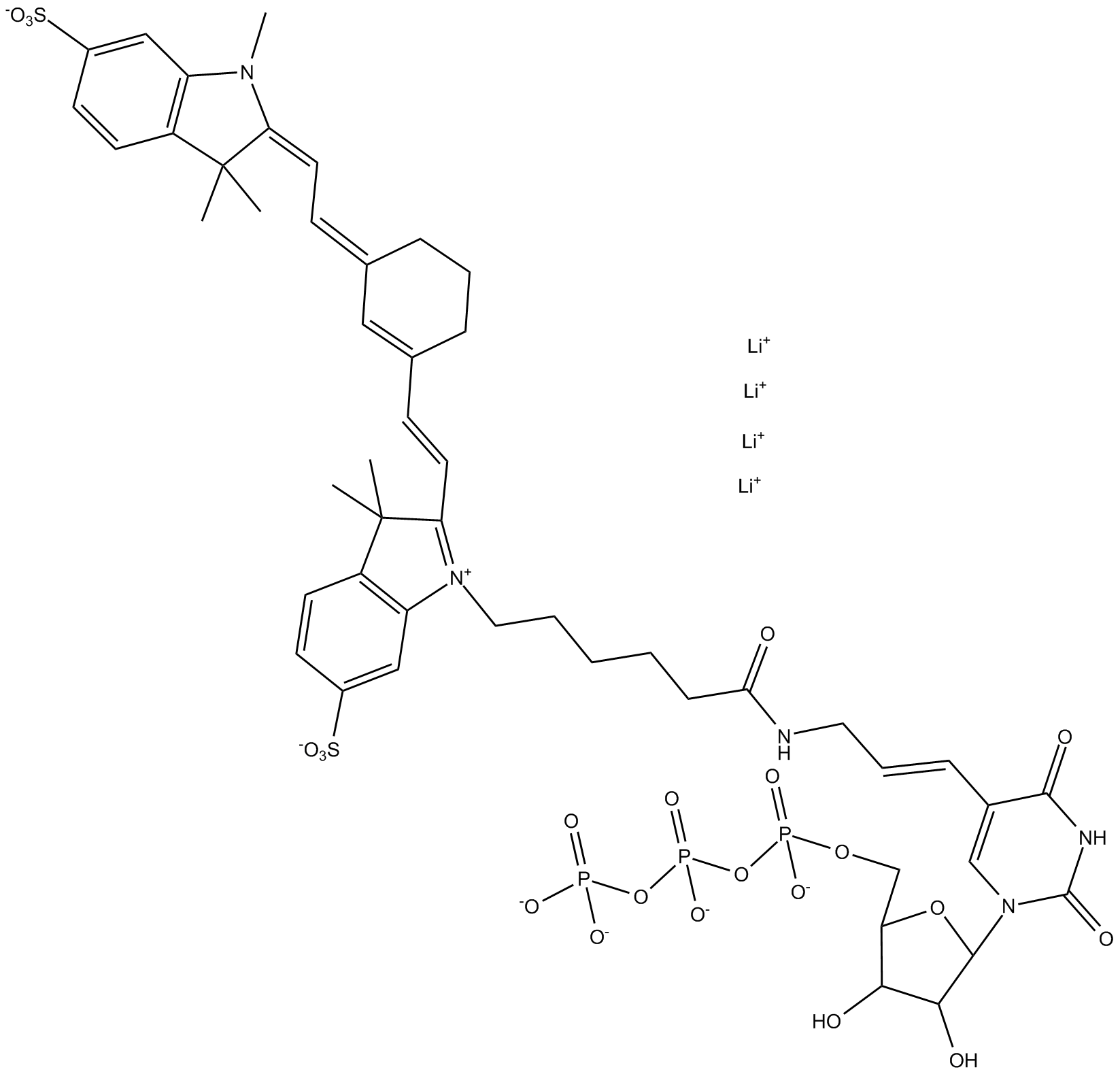 B8167 Cyanine 7-AA-UTP
B8167 Cyanine 7-AA-UTP -
 A8020 Cyanine 3 TyramideSummary: Cyanine 3 Tyramide
A8020 Cyanine 3 TyramideSummary: Cyanine 3 Tyramide -
 C8596 Cyanine 5.5 amineSummary: Cyanine5.5 amine can react with reactive groups such as NHS esters, carboxyl groups (after activation with carbodiimide), and epoxides.
C8596 Cyanine 5.5 amineSummary: Cyanine5.5 amine can react with reactive groups such as NHS esters, carboxyl groups (after activation with carbodiimide), and epoxides. -
 C8597 Cyanine 5 amine hydrochlorideSummary: Cy5 amine hydrochloride can react with reactive groups such as NHS esters, carboxyl groups (after activation with carbodiimide), and epoxides.
C8597 Cyanine 5 amine hydrochlorideSummary: Cy5 amine hydrochloride can react with reactive groups such as NHS esters, carboxyl groups (after activation with carbodiimide), and epoxides.


