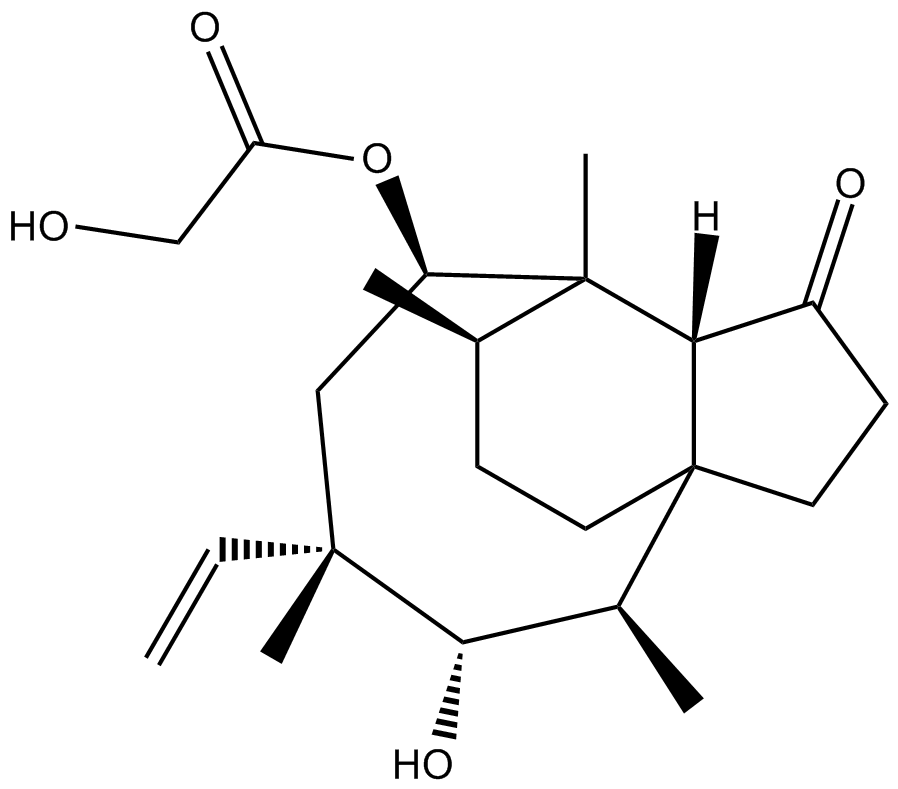
Pleuromutilin
Pleuromutilin is a diterpene antibiotic derived from the basidiomycete fungus, Clitopilus passeckerianus [1][2].
Pleuromutilin is a biologically active compound produced by Clitopilus passeckerianus, Drosophila subatrata, Clitopilus scyphoides (Pleurotus mutilus), and several other species of the genus Clitopilus. Pleuromutilin is a tricyclic diterpene that against gram-positive bacteria, such as Staphylococcus aureus, Streptococcus haemolyticus, and Bacillus subtilis. tiamulin and valnemulin, the derivatives of pleuromutilin, are used in veterinary medicine to treat swine dysentery and enzootic pneumonia. A novel semisynthetic pleuromutilin, retapamulin, is active against Staphylococcus aureus and Streptococcus pyogenes and is the first pleuromutilin derivative formulated as a topical antibacterial agent for treating human skin infections [1].
Pleuromutilin is a diterpene antibiotic that inhibits bacterial protein synthesis by binding to bacterial ribosomes in the peptidyl transferase center of the 50S subunit and inhibiting peptide bond formation [2].
References:
[1]. Kilaru S, Collins CM, Hartley AJ, et al. Establishing molecular tools for genetic manipulation of the pleuromutilin-producing fungus Clitopilus passeckerianus. Appl Environ Microbiol. 2009 Nov;75(22):7196-204.
[2]. Long KS, Hansen LH, Jakobsen L, et al. Interaction of pleuromutilin derivatives with the ribosomal peptidyl transferase center. Antimicrob Agents Chemother. 2006 Apr;50(4):1458-62.
| Physical Appearance | A crystalline solid |
| Storage | Store at 4°C |
| M.Wt | 378.5 |
| Cas No. | 125-65-5 |
| Formula | C22H34O5 |
| Synonyms | Antibiotic A 40104C,Drosophilin B,NSC 121145 |
| Solubility | ≥90.6 mg/mL in DMSO; ≥103.4 mg/mL in EtOH; insoluble in H2O |
| Chemical Name | 2-hydroxy-acetic acid-(3aS,4R,5S,6S,8R,9R,9aR,10R)-6-ethenyldecahydro-5-hydroxy-4,6,9,10-tetramethyl-1-oxo-3a,9-propano-3aH-cyclopentacycloocten-8-yl ester |
| SDF | Download SDF |
| Canonical SMILES | C[C@H](CCC(CC1)([C@H]2C1=O)[C@@H](C)[C@@H]([C@@](C)(C1)C=C)O)C2(C)[C@@H]1OC(CO)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
E. coli strains |
|
Preparation method |
The solubility of this compound in DMSO is ≤ 25mg/ml. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
200~250 μg/ml, >400 μg/ml |
|
Applications |
Relative to the parent strain, the L3 mutant strain had reduced sensitivity to tiamulin, pleuromutilin, and SB-264128. In contrast, no difference in susceptibility to valnemulin was observed between the parent and the L3 mutant strains. This is important, as mutations in the corresponding position of ribosomal protein L3 in the Brachyspira mutant screened in the laboratory and B. hyodysenteriae field isolates resulted in reduced sensitivity to tiamulin. |
|
References: [1]. Long KS, Hansen LH, Jakobsen L, Vester B. Interaction of pleuromutilin derivatives with the ribosomal peptidyl transferase center. Antimicrob Agents Chemother. 2006 Apr;50(4):1458-62. doi: 10.1128/AAC.50.4.1458-1462.2006. PMID: 16569865; PMCID: PMC1426994. |
|
Quality Control & MSDS
- View current batch:
Chemical structure