GMX1778 (CHS828)
GMX1778 (CHS828) is a potent and specific inhibitor of nicotinamide phosphoribosyltransferase (NAMPT) with a Kd value of 120 nM [1].
NAMPT is an NAD+ biosynthesis enzyme. NAD+ is a cofactor of enzymatic redox reactions that are involved in cellular metabolism, including ATP production. NAD+ is also important in many cellular pathways responsible for gene regulation, calcium homeostasis, genomic integrity, longevity and apoptosis. Cancer cells are significantly dependent on NAD+ for supporting high levels of ATP production for rapid cell proliferation [1].
Treatment with 30 nM GMX1778 for 6 h, extracts of IM-9 cells showed decreased NAD+ and NM levels, compared to cytosolic extracts of untreated cells. These levels continued to decrease throughout the rest of the time course experiment. The most profoundly changed was the metabolite NAD+ level. Between the 6- and 20-h time points, the decline of NAD+ occurred [1].
The NAD+ salvage pathway produces NAD+ using either nicotinic acid (niacin) (NA) or nicotinamide (niacinamide) (NM) as a substrate. NAPRT1 is short for NA phosphoribosyltransferase 1. On xenograft tumors derived from NAPRT1-proficient (HCT-116) and NAPRT1-deficient (HT1080) cell lines in mice, a 24-h iv infusion of GMX1777 at 150 mg/kg or 650 mg/kg produced antitumor activity. A 4-h iv infusion of NA at 120 mg/kg did not adversely affect the antitumor activity of GMX1777 in the NAPRT1-deficient xenograft, but abolished the antitumor activity of GMX1777 against the NAPRT1-proficient HCT-116 cell line [1].
Reference:
[1]. Watson M, Roulston A, Bélec L, et al. The small molecule GMX1778 is a potent inhibitor of NAD+ biosynthesis: strategy for enhanced therapy in nicotinic acid phosphoribosyltransferase 1-deficient tumors. Molecular and cellular biology, 2009, 29(21): 5872-5888.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 371.86 |
| Cas No. | 200484-11-3 |
| Formula | C19H22ClN5O |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥18.3 mg/mL in DMSO |
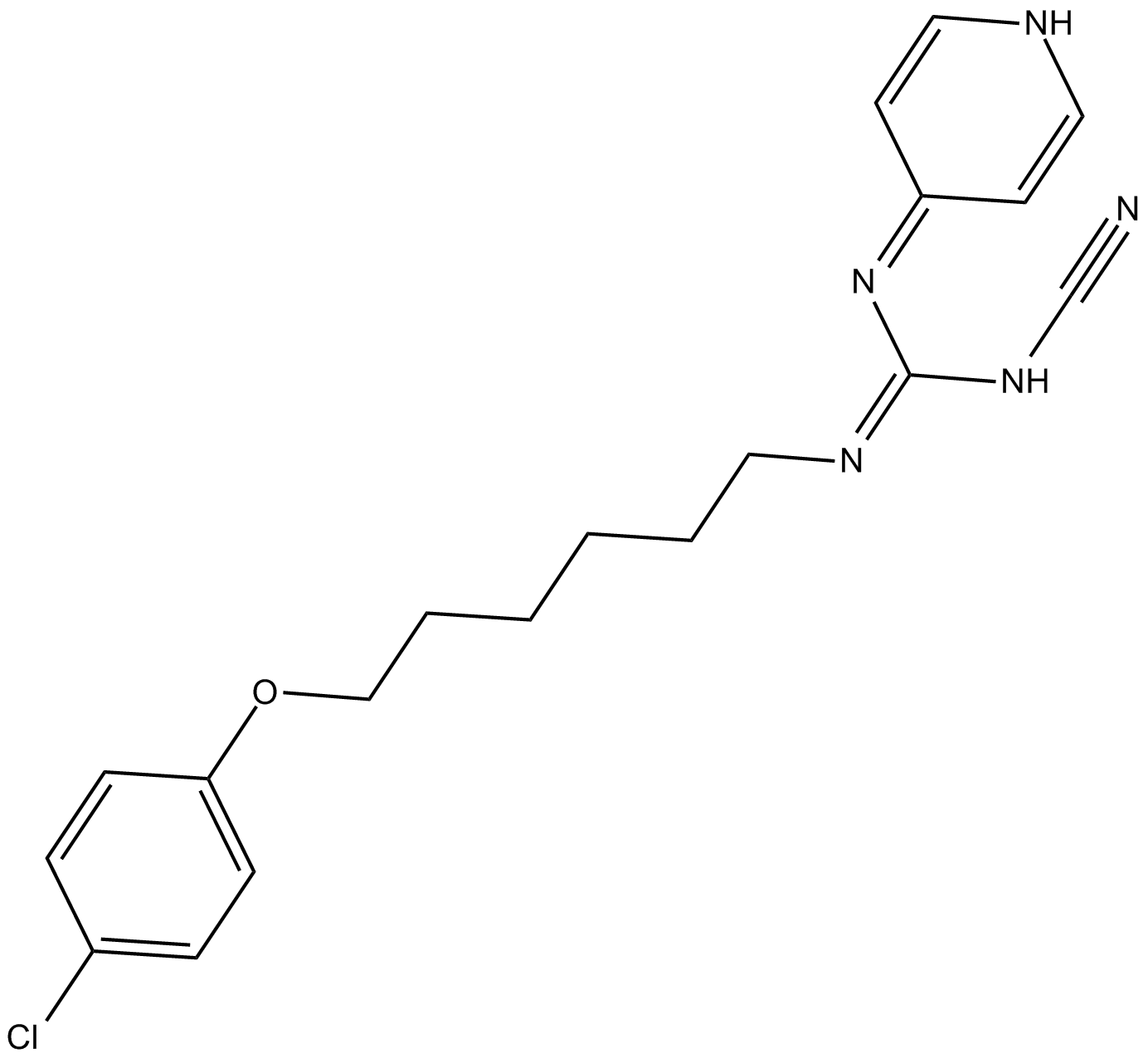
| Chemical Name | (Z)-2-(6-(4-chlorophenoxy)hexyl)-1-cyano-3-(pyridin-4(1H)-ylidene)guanidine |
| SDF | Download SDF |
| Canonical SMILES | N#CN/C(\N=C1C=CNC=C1)=N/CCCCCCOc(cc1)ccc1Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment:[1] | |
|
Cell lines |
IM-9 cells |
|
Reaction Conditions |
30 nM GMX1778 for 6 h incubation |
|
Applications |
Treatment with 30 nM GMX1778 for 6 h decreased NAD+ and nicotinamide (NM) levels in IM-9 cells, which continued to decrease throughout the rest of the time course experiment. The metabolite NAD+ level was found to be the most profoundly changed, and the NAD+ decline occurred between the 6- and 20-h time points. |
| Animal experiment:[2] | |
|
Animal models |
Nude mice bearing midgut carcinoid (GOT1), pancreatic carcinoid (BON) and medullary thyroid cancer (GOT2) tumors |
|
Dosage form |
100 or 250 mg/kg/week Administered orally (p.o,) |
|
Applications |
GMX1778 (250 mg/kg, p.o.) showed marked antitumor activity against three different human neuroendocrine tumors transplanted in nude mice. Histological examination of all three tumor types showed that in the high-dose group, necrosis was the predominant mode of cell death, although apoptotic cells were also found. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Watson M, Roulston A, Bélec L, et al. The small molecule GMX1778 is a potent inhibitor of NAD+ biosynthesis: strategy for enhanced therapy in nicotinic acid phosphoribosyltransferase 1-deficient tumors. Molecular and Cellular Biology, 2009, 29(21): 5872-5888. 2. Johanson V, Arvidsson Y, Kölby L, et al. Antitumoural effects of the pyridyl cyanoguanidine CHS 828 on three different types of neuroendocrine tumours xenografted to nude mice. Neuroendocrinology, 2005, 82(3-4): 171-176. |
|
Quality Control & MSDS
- View current batch:
Chemical structure