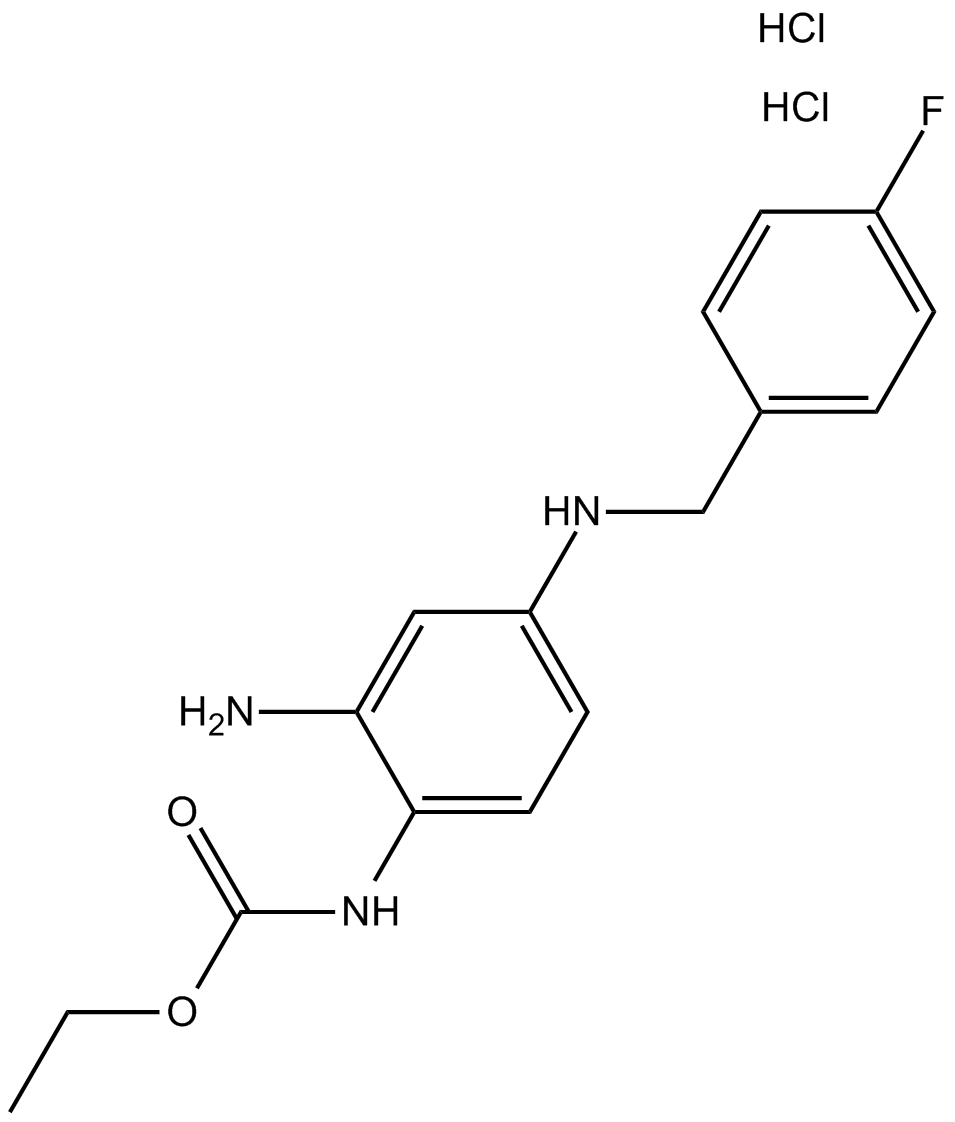
Retigabine dihydrochloride
IC50: N/A
Retigabine is a first-in-class K+ channel (KCNQ) opener. KCNQ channels are reported to be expressed predominantly in neurons and are critical determinants of cellular excitability, as shown by the occurrence of human genetic mutations in KCNQ channels which underlie inheritable disorders including the syndrome of benign familial neonatal convulsions.
In vitro: Retigabine was found to combine a novel mode of actions, which were namely potassium channel opening (KCNQ2, KCNQ3 as well as KCNQ4 channels). Retigabine also showed activities with some potentiation of gamma amino butyric acid (GABA)-evoked currents at its higher concentrations [1].
In vivo: Animal models of epileptic seizures showed that retigabine treatment was effective at an oral dose as low as 0.01 mg/kg. Studies performed in mice also indicated that combining retigabine with another anticonvulsant agent leads to an additive effect [1].
Clinical trial: A clinical interaction study showed that there was no pharmacokinetic interaction between phenobarbitone and retigabine in healthy subjects. Thus, no dosage adjustment is likely to be necessary when phenobarbitone and retigabine are coadministered to patients [1].
Reference:
[1] Ferron GM,Patat A,Parks V,Rolan P,Troy SM. Lack of pharmacokinetic interaction between retigabine and phenobarbitone at steady-state in healthy subjects. Br J Clin Pharmacol.2003 Jul;56(1):39-45.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 376.25 |
| Cas No. | 150812-13-8 |
| Formula | C16H20Cl2FN3O2 |
| Synonyms | D 20443 dihydrochloride;D-20443 dihydrochloride;D20443 dihydrochloride |
| Solubility | ≥18.8 mg/mL in DMSO; ≥52.4 mg/mL in H2O with gentle warming; ≥8.71 mg/mL in EtOH with gentle warming and ultrasonic |
| Chemical Name | ethyl N-[2-amino-4-[(4-fluorophenyl)methylamino]phenyl]carbamate;dihydrochloride |
| Canonical SMILES | CCOC(Nc(ccc(NCc(cc1)ccc1F)c1)c1N)=O.Cl.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
Chinese hamster ovary (CHO-K1) cells transfected with the KCNQ2/Q3 tandem construct (CHO-KCNQ2/Q3) |
|
Preparation method |
This compound is soluble in DMSO. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reaction Conditions |
0.1 ~ 10 μM |
|
Applications |
Retigabine Dihydrochloride (0.1 ~ 10 μM) induced a potassium current and hyperpolarized CHO-KCNQ2/Q3 cells but not in wild-type cells. |
| Animal experiment [2]: | |
|
Animal models |
Mouse 6-Hz psychomotor seizure models |
|
Dosage form |
26 or 33 mg/kg; i.p. |
|
Applications |
In the 6-Hz psychomotor seizure model, Retigabine Dihydrochloride dose-dependently blocked seizures induced by either 32 or 44 mA current stimulation. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Wickenden AD, Yu W, Zou A, Jegla T, Wagoner PK. Retigabine, a novel anti-convulsant, enhances activation of KCNQ2/Q3 potassium channels. Mol Pharmacol. 2000 Sep;58(3):591-600. [2]. Large CH, Sokal DM, Nehlig A, Gunthorpe MJ, Sankar R, Crean CS, Vanlandingham KE, White HS. The spectrum of anticonvulsant efficacy of retigabine (ezogabine) in animal models: implications for clinical use. Epilepsia. 2012 Mar;53(3):425-36. | |
Quality Control & MSDS
- View current batch:
Chemical structure